CE / CME
Choosing Optimal Second-line Therapy for PBC
Pharmacists: 0.75 contact hour (0.075 CEUs)
Physicians: maximum of 0.75 AMA PRA Category 1 Credit™
Nurse Practitioners/Nurses: 0.75 Nursing contact hour
Released: March 31, 2026
Expiration: March 30, 2027
Activity
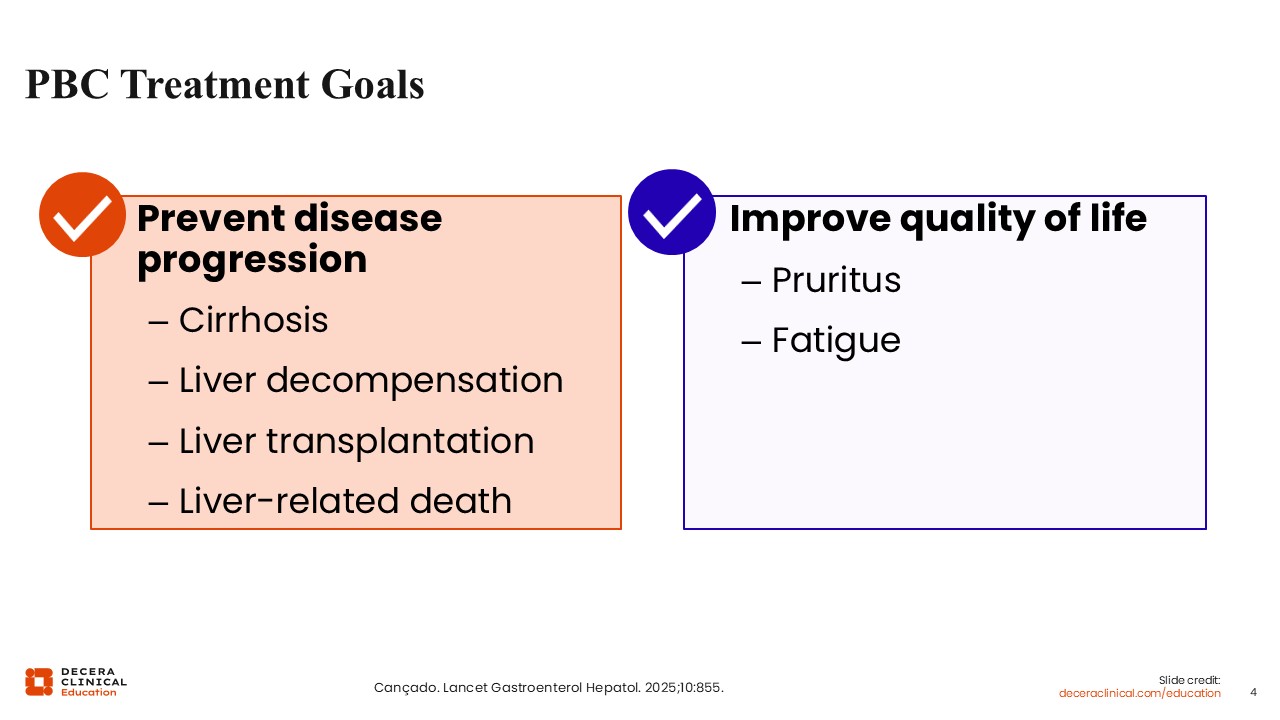
PBC Treatment Goals
Today, we discuss choosing optimal second-line therapy for people living with PBC. This is important because our goal is to help all people living with PBC achieve the best possible disease control and quality of life.
Because PBC is a chronic liver disease, untreated or inadequately treated disease can progress to cirrhosis. Once cirrhosis develops, patients are at risk for liver decompensation, liver transplantation, and liver-related death.1
The key symptoms of PBC are pruritus and fatigue, both of which are highly bothersome to patients and should be considered part of the overall treatment goal.1
PBC Treatment Goals
Today, we discuss choosing optimal second-line therapy for people living with PBC. This is important because our goal is to help all people living with PBC achieve the best possible disease control and quality of life.
Because PBC is a chronic liver disease, untreated or inadequately treated disease can progress to cirrhosis. Once cirrhosis develops, patients are at risk for liver decompensation, liver transplantation, and liver-related death.1
The key symptoms of PBC are pruritus and fatigue, both of which are highly bothersome to patients and should be considered part of the overall treatment goal.1
When to Advance to Second-line Treatment: Baseline Risk Factors for Disease Progression/Treatment Failure
Baseline risk factors in PBC include features identified in cohort studies as being associated with increased risk of disease progression to liver failure, death, or liver transplantation, as well as a greater likelihood of treatment failure with UDCA.4-6
These include advanced fibrosis, which is now more commonly assessed by liver stiffness rather than liver biopsy. Higher baseline ALP and bilirubin are also associated with a less adequate response to UDCA.7,8
Similarly, ductopenia is more prevalent in severe PBC and serves as an independent risk factor for treatment failure and negative disease outcomes.9 Younger age is a key risk factor: the younger the patient at diagnosis, the greater the likelihood of insufficient response to UDCA. Younger people are also often more symptomatic and may be more likely to have ductopenia and/or greater inflammatory activity.10
Finally, although PBC is most predominant in women, male gender has been associated with UDCA nonresponse.10on average, less than 50% of people diagnosed at 50 years or younger will be sufficient UDCA responders.
Finally, there is some evidence to support that men have a higher risk of disease progression and insufficient response to UDCA.
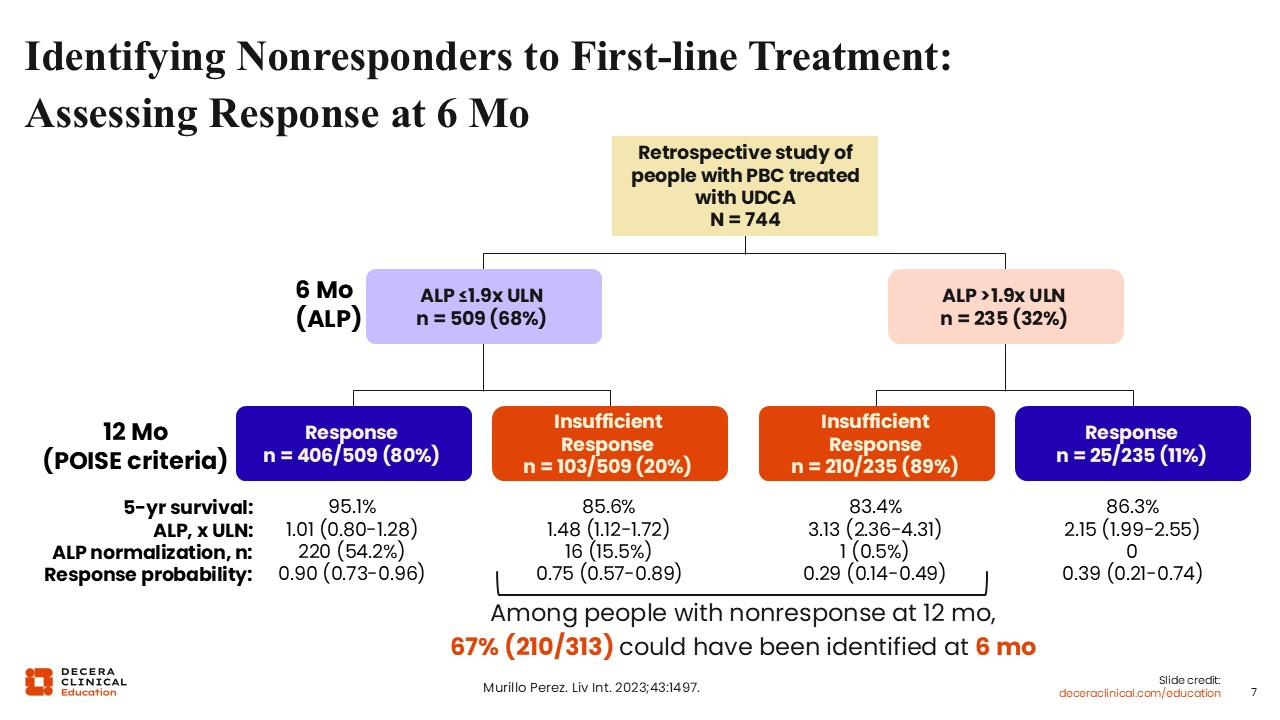
Identifying Nonresponders to First-line Treatment: Assessing Response at 6 Months
There are data showing that the classical 12-month assessment of biochemical response to UDCA can be moved forward to 6 months. This was demonstrated in a global PBC cohort study using retrospective data that initially examined ALP at 6 months rather than waiting until 12 months. The analysis showed that most people who ultimately would be classified as nonresponders at 12 months using the historic POISE criteria often could be identified earlier.11
This work is best viewed as proof of concept that assessment can reasonably be performed at 6 months rather than 12 months. At the same time, our treatment goals have continued to evolve. Although the POISE criteria remain important, healthcare professionals are now aiming for more ambitious outcomes, including ALP normalization.
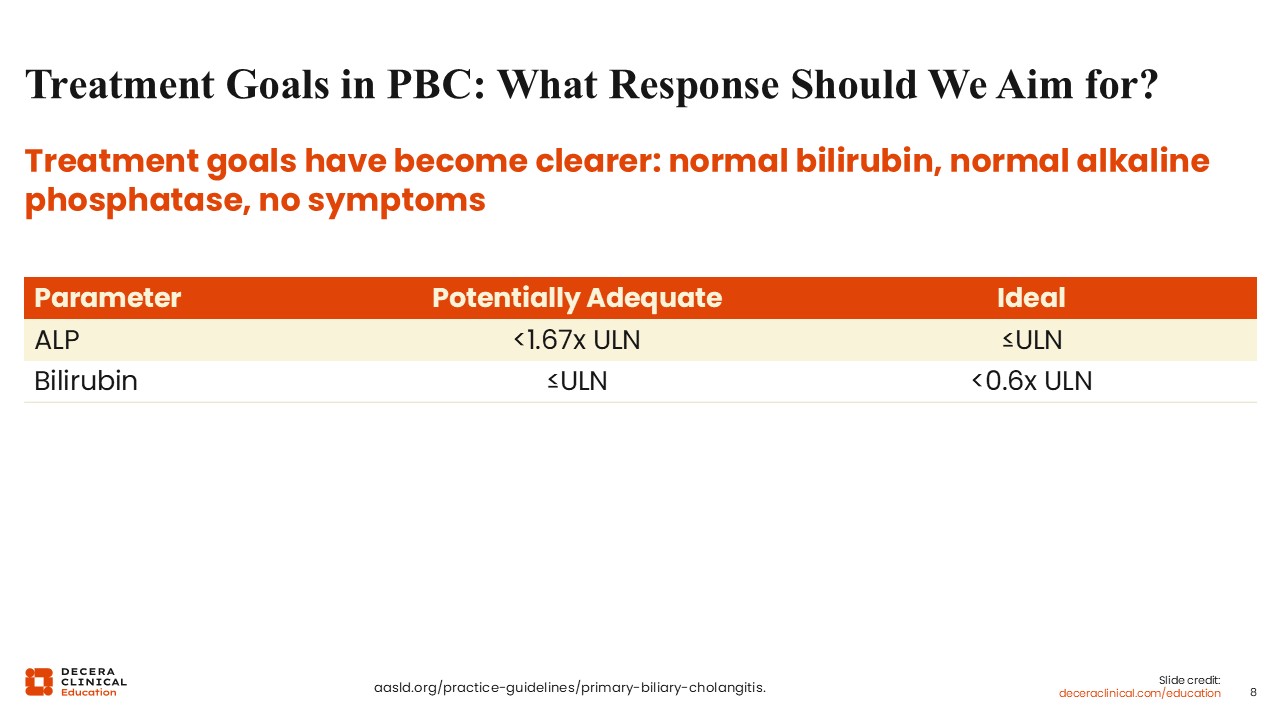
Treatment Goals in PBC: What Response Should We Aim for?
What response should we aim for when treating PBC? To keep this simple, I believe healthcare professionals should focus on 3 broad goals: normalizing bilirubin, normalizing ALP, and resolving symptoms.
An adequate treatment response might be considered an ALP level less than 1.67 times the upper limit of normal with a bilirubin below the upper limit of normal. However, an ideal response would be normalization of ALP and bilirubin less than 0.6 times the upper limit of normal.3,12
However, these goals still need to be individualized according to the patient’s overall risk profile.
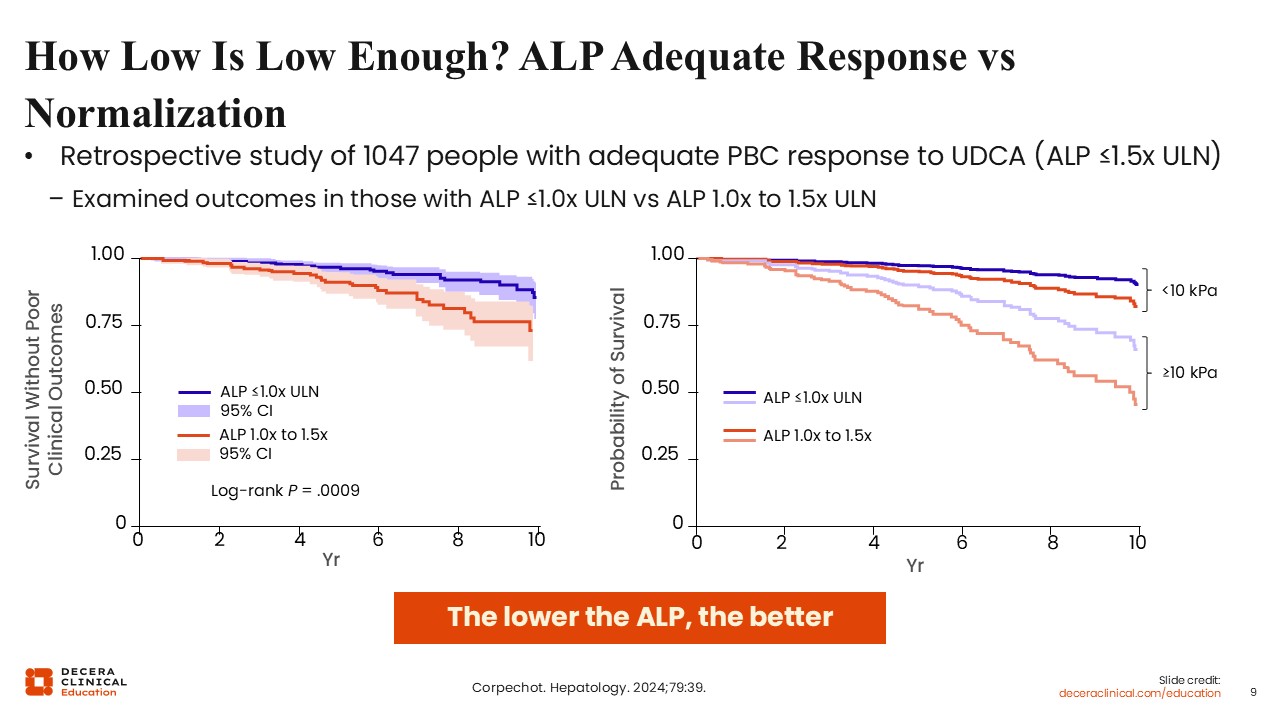
How Low Is Low Enough? ALP Adequate Response vs Normalization
The distinction between adequate and ideal treatment goals is data driven. Global PBC data have shown that, even within the normal range, bilirubin predicts outcome in PBC, and that among people with normal bilirubin, those with normal ALP have the most favorable outcomes.13
Another global PBC analysis further demonstrated that people with ALP less than the upper limit of normal have the best outcomes. When liver stiffness is added, risk can be stratified further. Even so, regardless of liver stiffness, lower ALP remains beneficial. The most favorable outcomes are seen in people with liver stiffness <10 kPa and a normal ALP.14
The practical message is straightforward: the lower the ALP, the better. The target should be normalization whenever possible.3
Second-line Treatment of PBC
What can we do in the second-line treatment landscape for PBC? Around the world, off-label fibric acid derivatives, or fibrates, have been used for decades. Obeticholic acid was the other historical second-line treatment option, but it was withdrawn from the US market in September 2025 and is no longer available.2,12,15,16
Two next-generation PPAR agonists received FDA approval in 2024. Elafibranor, dosed at 80 mg, is a next-generation PPAR-alpha/delta agonist. Seladelpar, dosed at 10 mg, is a next-generation PPAR-delta agonist. Both are approved in combination with UDCA for adults with an inadequate response to UDCA or as monotherapy for those who are intolerant of UDCA.17,18
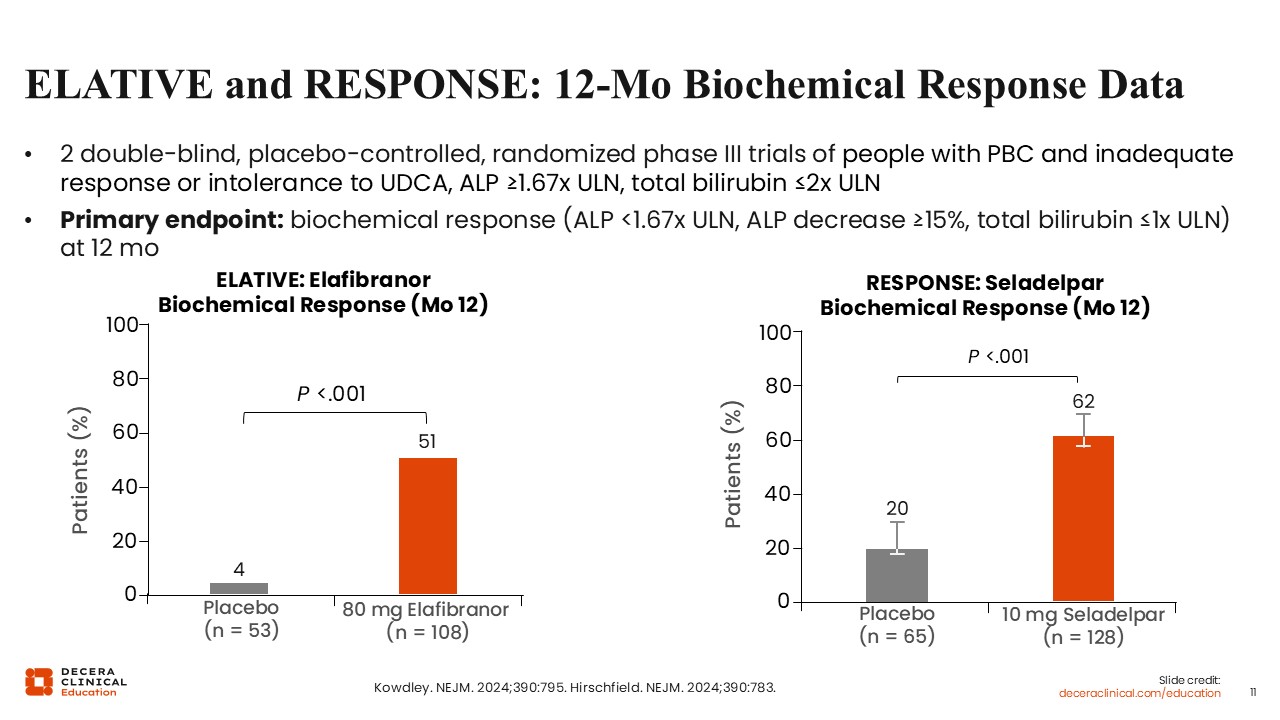
ELATIVE and RESPONSE RCTs: 12-Month Biochemical Response Data
It is helpful to revisit the 2 pivotal phase III trials that supported the FDA approval of the next-generation PPAR agonists. The study populations were similar, and each trial randomized participants to a next-generation PPAR agonist or placebo over a 12-month double-blind period. In ELATIVE, they received elafibranor 80 mg, and in RESPONSE, they received seladelpar 10 mg.19,20
The predefined primary endpoint in both trials was biochemical response, defined as an ALP level <1.67 times the upper limit of normal, a reduction of at least 15% from baseline, and a normal total bilirubin level.19,20
In ELATIVE, among 161 total participants, biochemical response at week 52 was observed in 51% receiving elafibranor vs 4% receiving placebo.19
In RESPONSE, among 193 participants, biochemical response at Week 52 was observed in 62% receiving seladelpar vs 20% receiving placebo.20
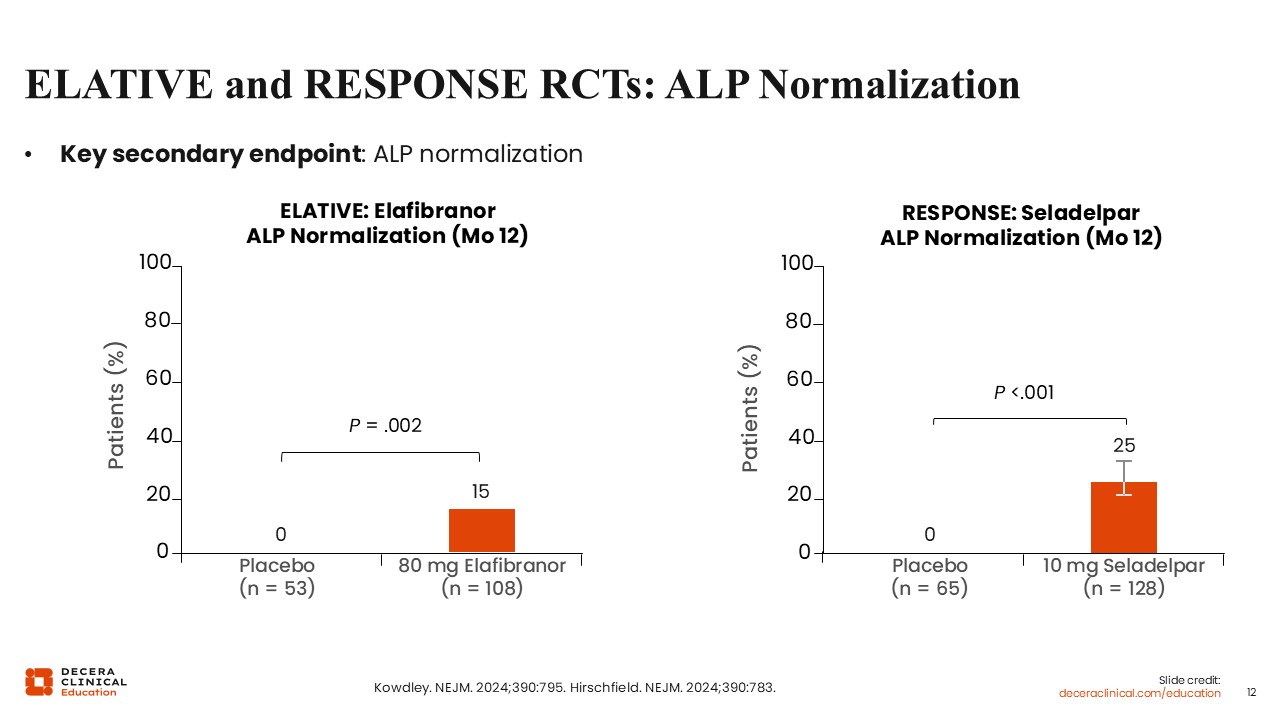
ELATIVE and RESPONSE RCTs: ALP Normalization
ALP normalization at Month 12 was a key secondary endpoint in both trials.
In the ELATIVE trial, it occurred in 15% of participants receiving elafibranor and in none receiving placebo.
In the RESPONSE trial, it occurred in 25% of participants receiving seladelpar and in none receiving placebo.19,20
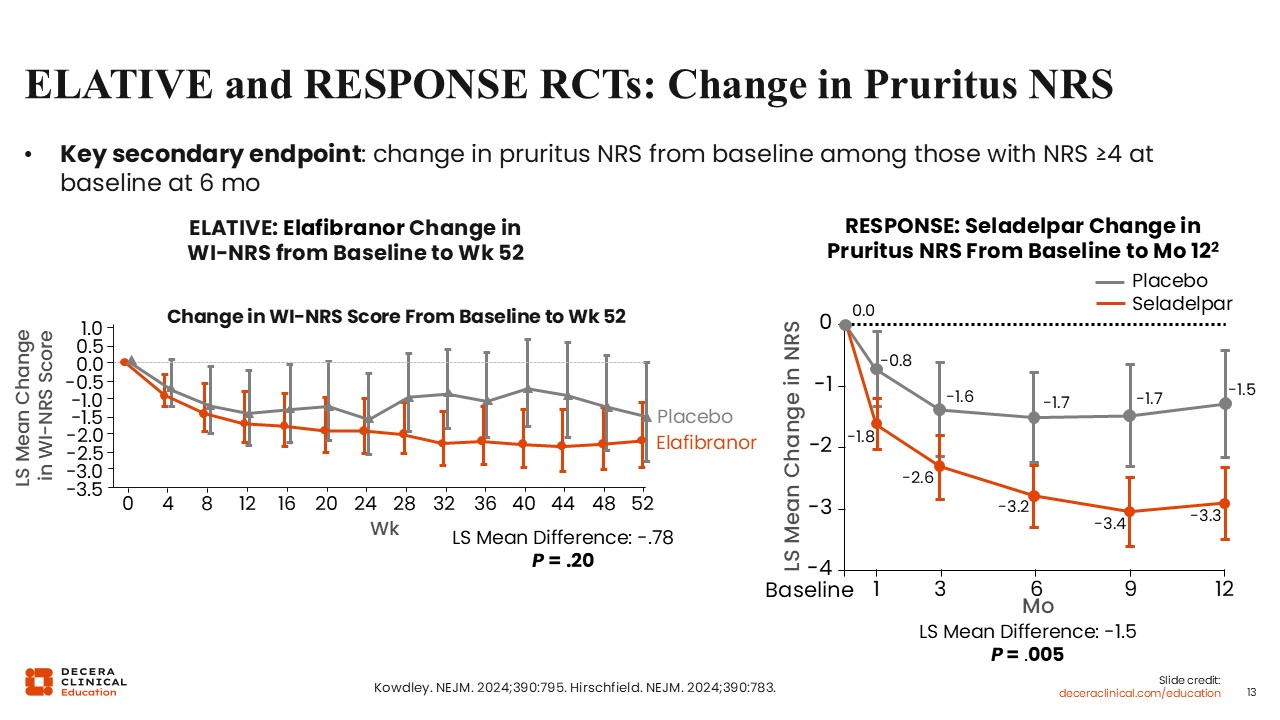
ELATIVE and RESPONSE RCTs: Change in Pruritus NRS
Change in pruritus symptoms, as characterized by the worst itch numeric rating scale, was another key secondary endpoint among participants with moderate to severe pruritus at baseline.
Among participants enrolled on the ELATIVE trial, the least squares mean change in worst itch from baseline did not differ significantly between elafibranor and placebo at 6 months. Among participants enrolled on the RESPONSE trial, the least squares mean change in worst itch from baseline was statistically significant with a greater reduction with seladelpar compared with placebo at 6 months.19,20
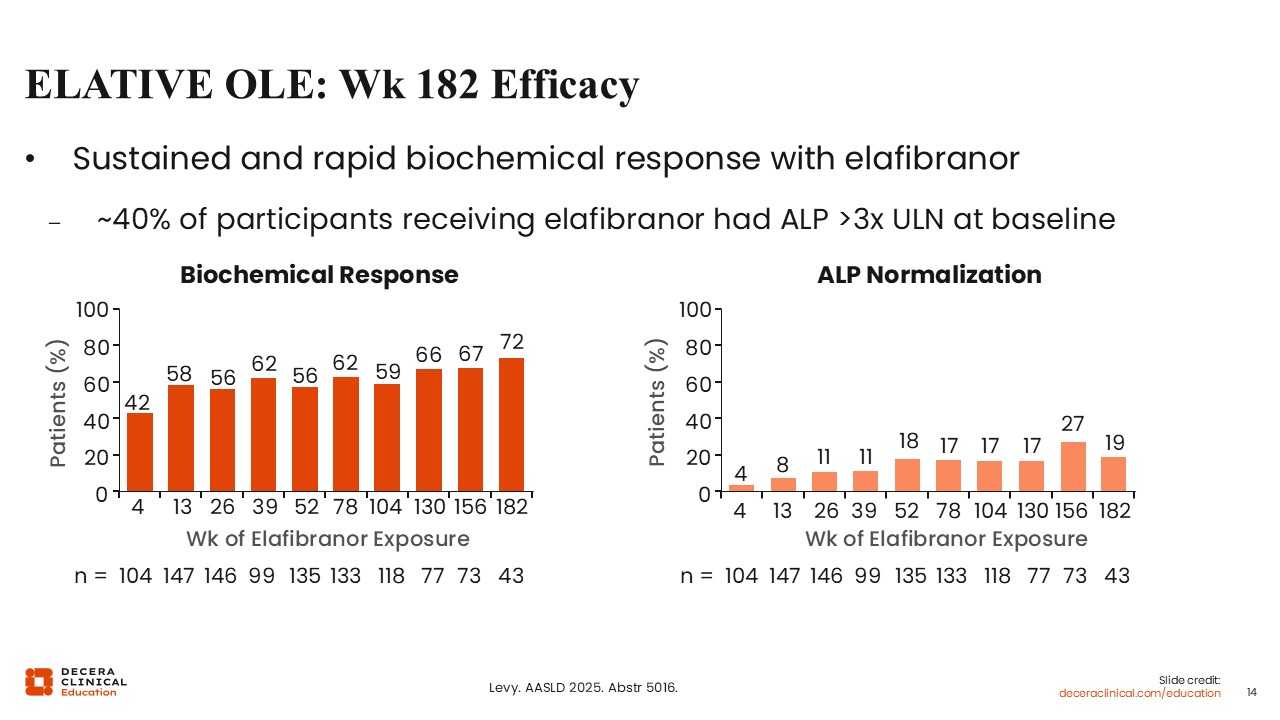
ELATIVE OLE: Week 182 Efficacy
We now even have additional data from the open-label extension studies of these next-generation PPAR agonists, reinforcing their efficacy as second-line therapy. For elafibranor, the ELATIVE open-label extension through 182 weeks was presented at a recent conference.21
These data demonstrated the long-term efficacy of elafibranor: The percentage of participants with composite biochemical response remained in the range of approximately 66% to 72% throughout follow-up, whereas the percentage with ALP normalization remained approximately 17% to 19% throughout follow-up.21
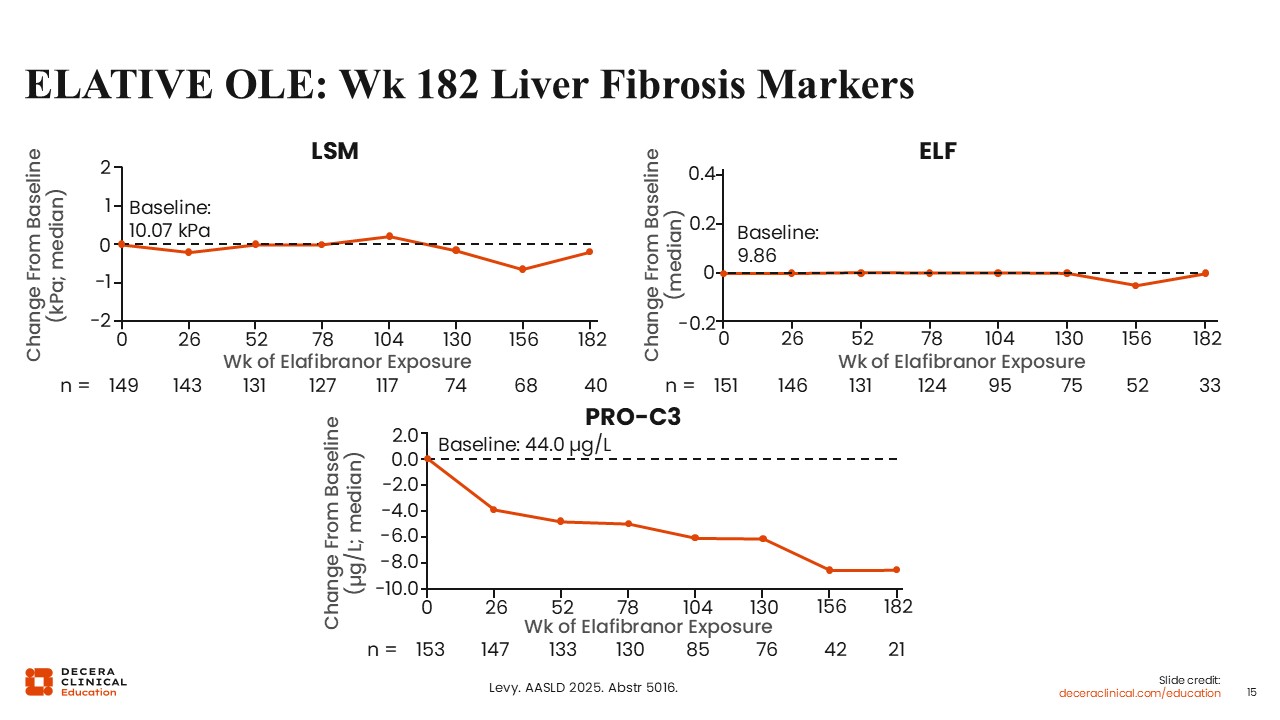
ELATIVE OLE: Week 182 Liver Fibrosis Markers
We also now have data on liver stiffness, enhanced liver fibrosis, and PRO-C3 from the elafibranor open-label extension. These markers provide additional information related to fibrosis, inflammation, and cholestasis.21
Across the extension period, liver stiffness remained stable and did not worsen with elafibranor treatment. Enhanced liver fibrosis also remained stable. In addition, PRO-C3 levels declined.21
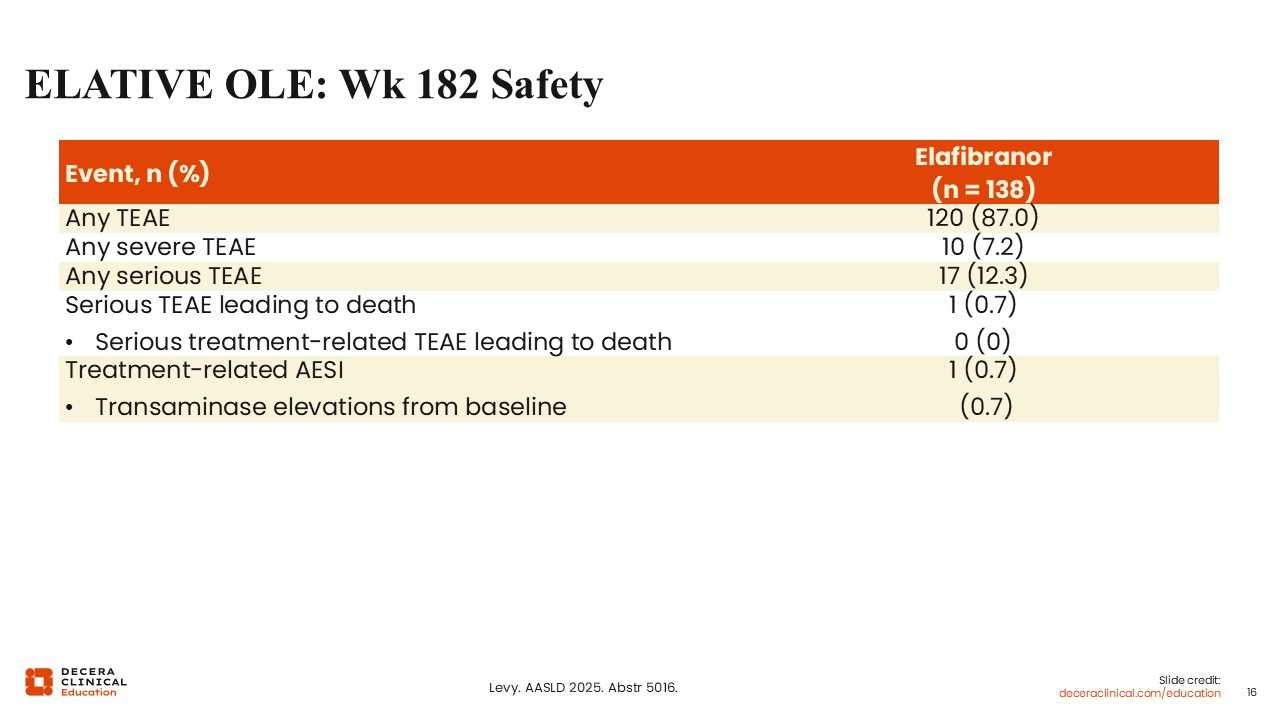
ELATIVE OLE: Week 182 Safety
Finally, no new safety signals were reported in the elafibranor open-label extension.21
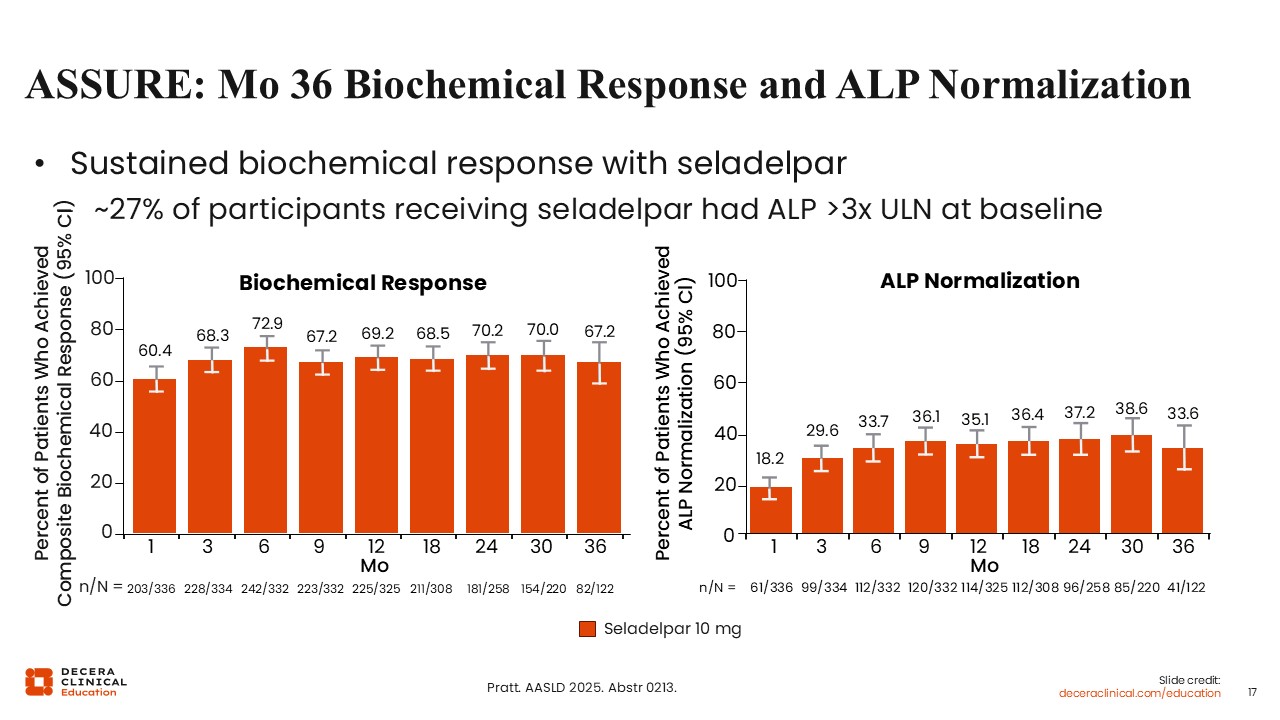
ASSURE: Month 36 Biochemical Response and ALP Normalization
The ASSURE study then provided an opportunity for participants from RESPONSE and other seladelpar studies to receive open-label seladelpar 10 mg daily.22
An interim analysis was presented in recent conference proceedings. Through 36 months, approximately 70% of participants achieved biochemical response. In addition, more than one third had achieved ALP normalization.22
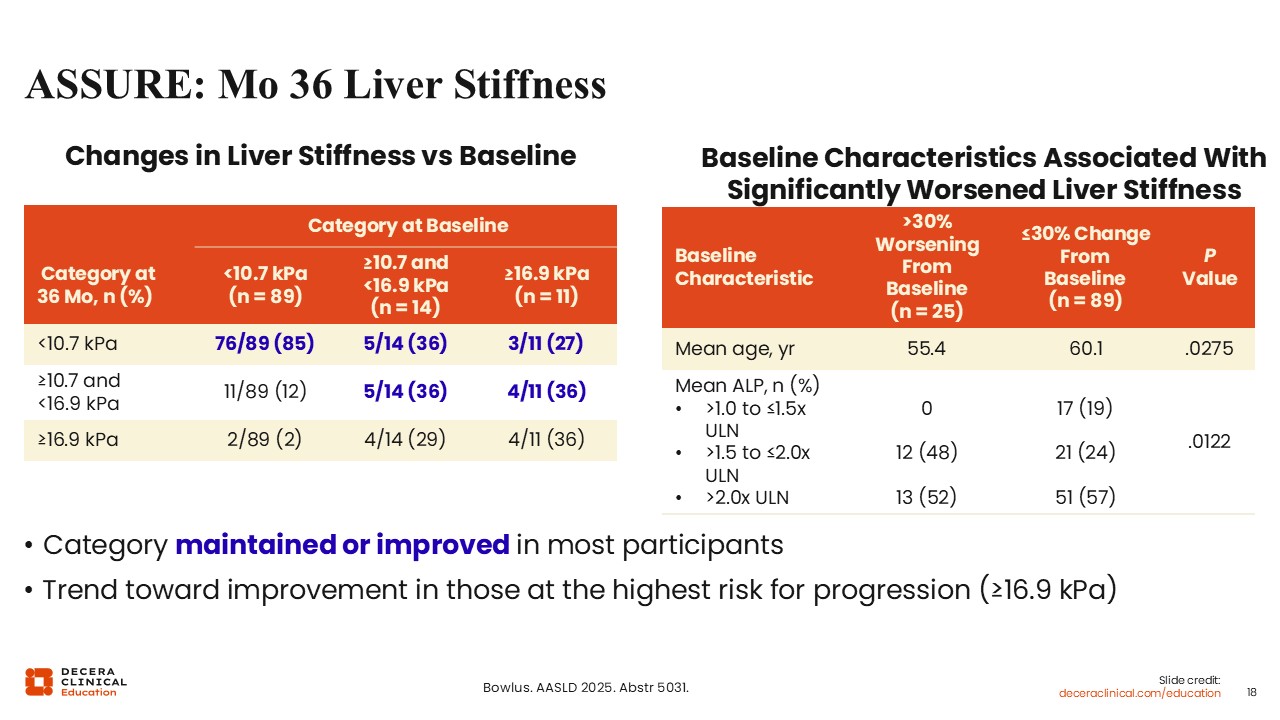
ASSURE: Month 36 Liver Stiffness
Liver stiffness data were also presented, showing maintenance or improvement for most participants receiving seladelpar.23
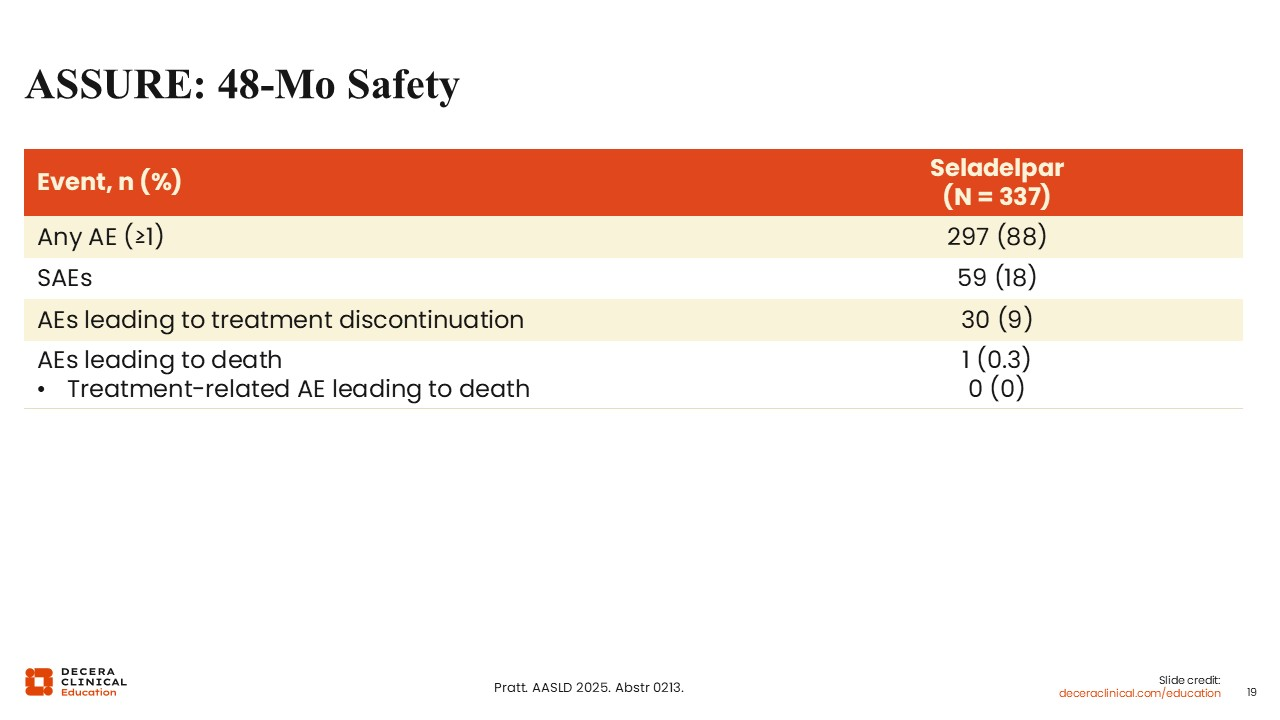
ASSURE: 48-Month Safety
As with elafibranor, no new safety signals were reported for seladelpar during the long-term open-label extension.22
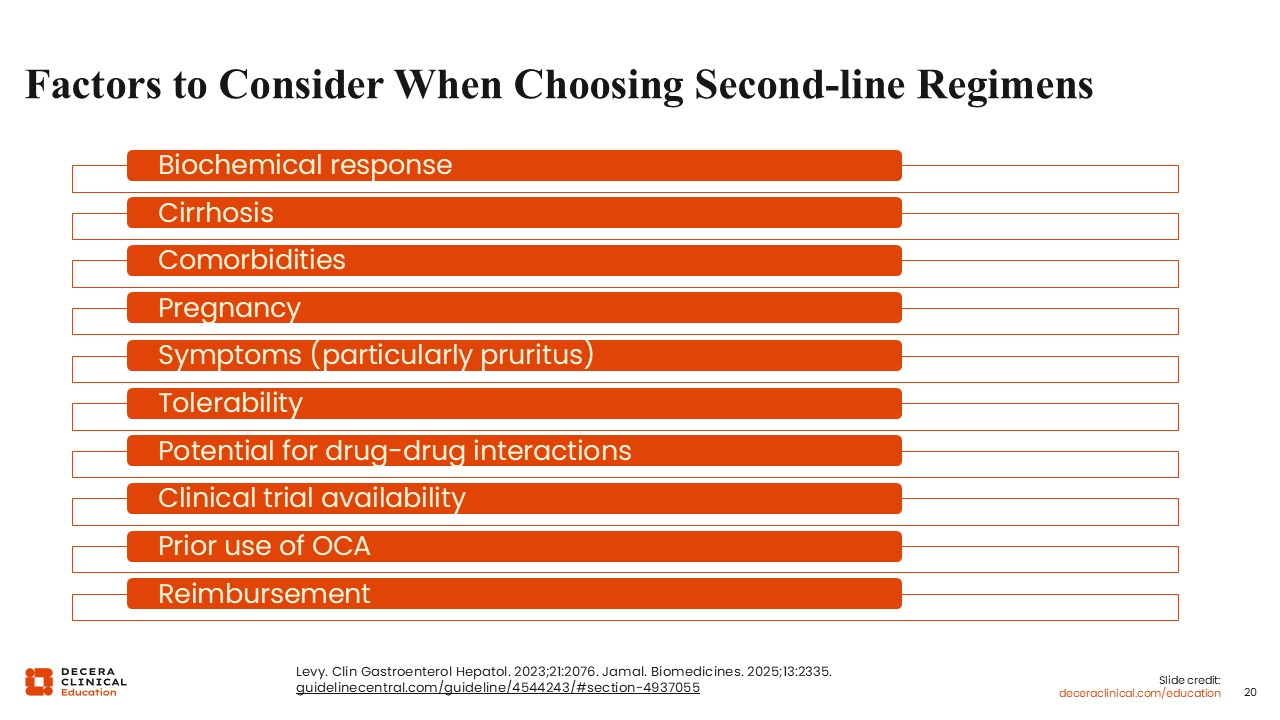
Factors to Consider When Choosing Second-line Regimens
To recap, the ideal treatment goals for second-line therapy in PBC are: normalize ALP, normalize bilirubin, and resolve symptoms. With those goals in mind, treatment selection should consider the patient’s current ALP and bilirubin, the presence of cirrhosis, comorbidities, age, and symptom burden, particularly pruritus.5,25,26
Additional considerations include tolerability and the potential for drug–drug interactions. For some people, future family planning may also be relevant. Reimbursement and access are practical considerations that can also influence treatment choice.5,25,26
Second-line PPAR Treatment of PBC: FDA Label Considerations
If we look specifically at the FDA labels for elafibranor and seladelpar, there are several points that healthcare professionals should keep in mind, including teratogenic potential with elafibranor.17,18 If a woman with reproductive potential does not want to take nonhormonal contraception, seladelpar should be used.
Second-line PPAR Treatment of PBC: FDA Label Drug-Drug Interactions
The FDA labels also include drug–drug interaction considerations for both elafibranor and seladelpar, as detailed on this slide.17,18
Second-line Treatment of PBC in People With Cirrhosis
In people with cirrhosis, treatment selection requires careful attention to risk and benefit.
In people with compensated cirrhosis, the potential benefit of second-line treatment remains clinically important, so they should still be considered for therapy.2,12,17,18
However, all of these agents are contraindicated or not recommended in Child-Pugh B or C cirrhosis.2,12,17,18
Key Points
In conclusion, next-generation PPAR agonists are associated with significant improvements in biochemical response and ALP normalization.
These remain major treatment goals in people living with PBC, alongside improvement in symptoms, particularly itch.