CE / CME
Clinical Focus in Hemophilia: Integrating Hemostatic Rebalancing Therapies Into Personalized, Evidence-Based Patient Care
ABIM MOC: maximum of 0.75 Medical Knowledge MOC point
Physicians: Maximum of 0.75 AMA PRA Category 1 Credit™
Released: March 16, 2026
Expiration: September 15, 2026
Activity
Hemophilia Disease Burden
Angela Weyand, MD:
Hemophilia A is a congenital X-linked disorder that is caused by a deficiency of factor VIII, whereas hemophilia B is caused by a deficiency of factor IX. Hemophilia A is estimated to affect approximately 1 in every 4000 live births in males. Hemophilia B is less common, affecting 1 in 20,000 live births in males. Unfortunately, the prevalence in women is less understood, as historically it was thought that women could not be affected. However, as more research is done, we now know that women can be affected. Severity has historically been defined by factor level, with patients with residual factor levels less than 1% of normal being classified as severe, those with residual factor levels between 1% and 5% are classified as moderate, and those with 6% to 40% are classified as having mild disease.1
Hemophilia Pathophysiology
Angela Weyand, MD:
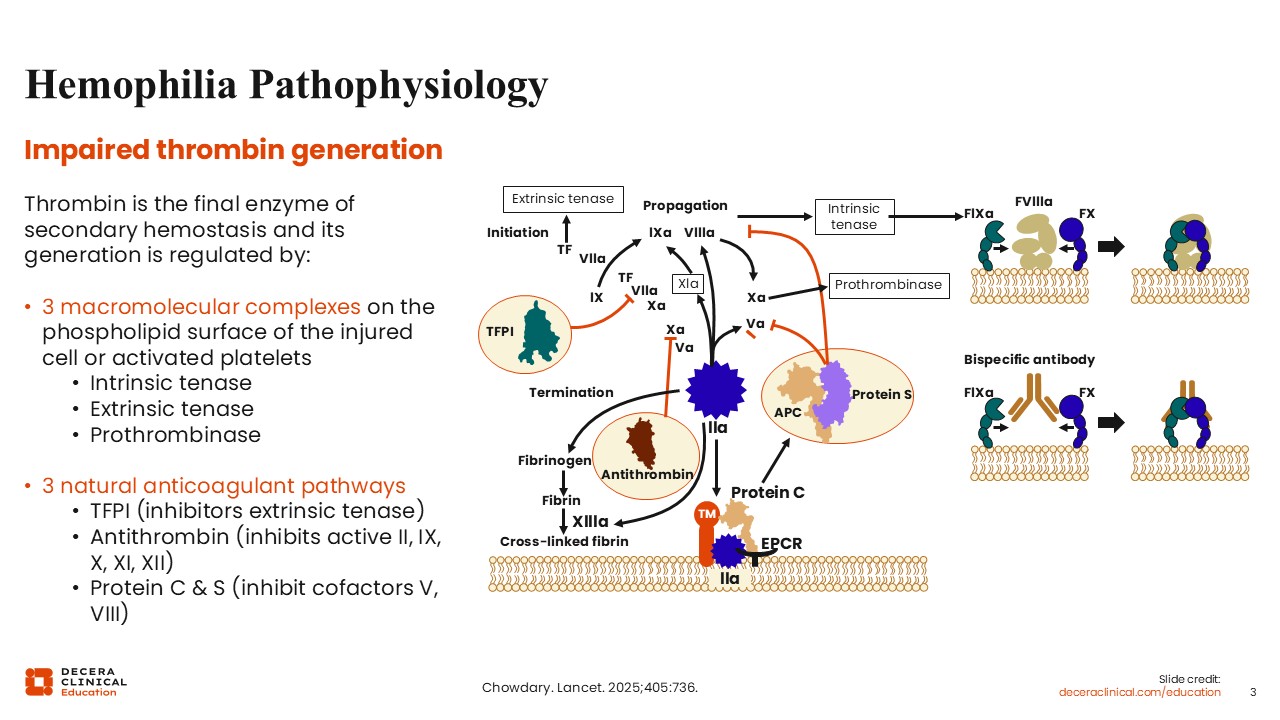
The problematic clinical issue is impaired thrombin generation. Thrombin is the final enzyme of secondary hemostasis, and its generation is regulated by 2 competing processes. The first process is comprised of 3 macromolecular complexes on the phospholipid surface of either injured cells or activated platelets. Procoagulants are grouped into 3 macromolecular complexes: the intrinsic tenase, the extrinsic tenase, and the prothrombinase. These procoagulant complexes contribute to hemostasis. The first process is balanced by the second process, which comprises 3 natural anticoagulant pathways. Tissue factor pathway inhibitor (TFPI) inhibits the extrinsic tenase antithrombin, which inhibits active factor II, factor IX, factor X, factor XI, and factor XII, and then protein C & S, which work together to inhibit the cofactors factor V and factor VIII. 1
Hemophilia Symptoms
Angela Weyand, MD:
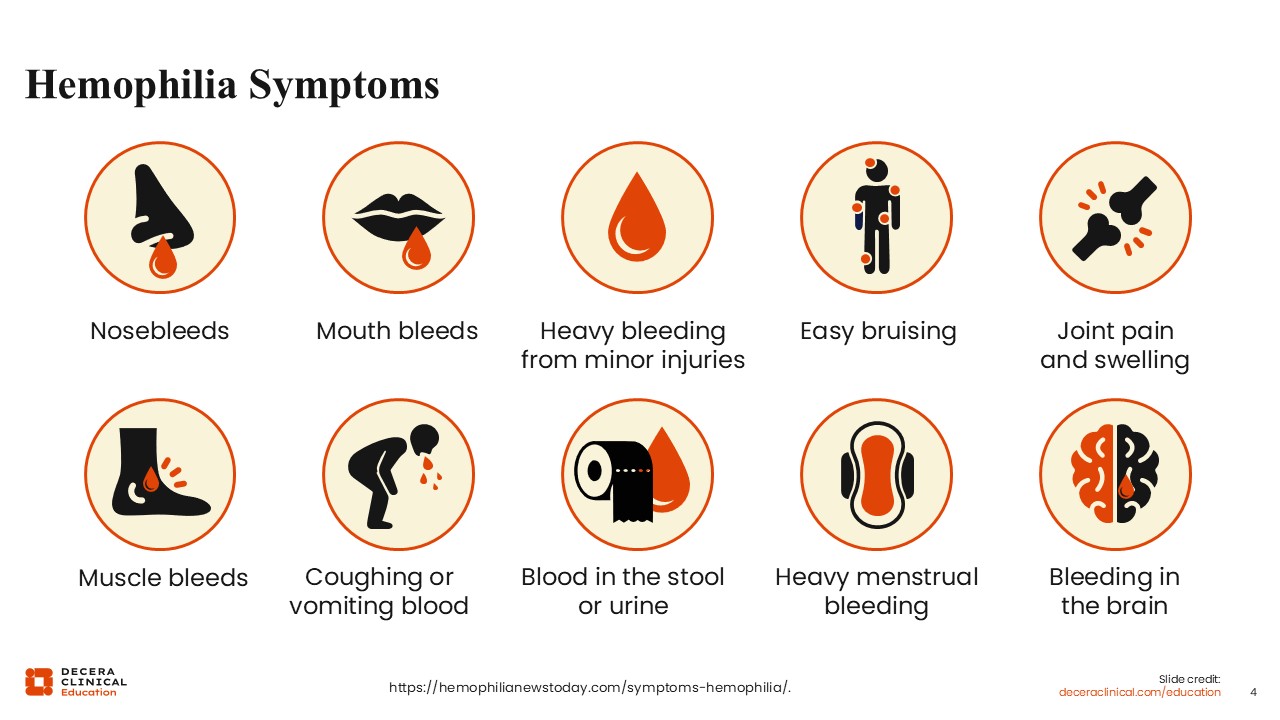
The symptoms of hemophilia are broad and can include mucosal nose bleeds, mouth bleeds, easy bruising, deeper bleeding and bruising, such as hemarthrosis or bleeding into the joint, and bleeding into muscles.
Bleeding into the joints can cause issues such as joint pain and swelling. Patients can also encounter more serious and sometimes life-threatening bleeding, such as bleeding into their brain or coughing or vomiting of blood.2
Patients With Hemophilia Continue to Have Unmet Needs
Angela Weyand, MD:
Despite a lot of progress in the care of patients with hemophilia, our patients continue to have unmet needs across the lifespan from neonates through adulthood. As they grow up and start to become more mobile, they encounter bleeding complications such as hemarthrosis.3 They often see signs of early joint damage and may have difficulty fitting in with peers due to hesitation to participate in certain activities. They may hesitate to participate in activities not only due to bleeding risk, but also due to the burden of treatment that exists with having to infuse factor or using other products for prophylaxis.4
During their early years, patients increase levels of physical activity, putting them at greater risk for bleeds.5 As they enter adolescence, they often experience challenges with adherence to their prescribed therapies, which can also predispose them to increased bleeding.
As patients enter adulthood, these earlier episodes of hemarthrosis, or even microbleeds that they may not have been clinically aware of, contribute to hemophilic arthropathy. This often is accompanied by chronic pain and a psychosocial burden, which manifests with an inability to attain the same quality of life as their peers without hemophilia, and dealing with comorbidities that often are at odds with their hemophilia treatment.6
As patients age, they may need treatments like blood thinners or antiplatelet agents. Our geriatric patients are at risk of devastating bleeding, such as intracranial hemorrhage. Patients with hemophilia still experience persistent bleeding, the joint damage and arthropathy that accompany that bleeding, and the chronic pain that results from bleeding.7 All these factors contribute overall to an impaired quality of life.
Patients Continue to Experience Bleeds Regardless of Disease Severity
Angela Weyand, MD:
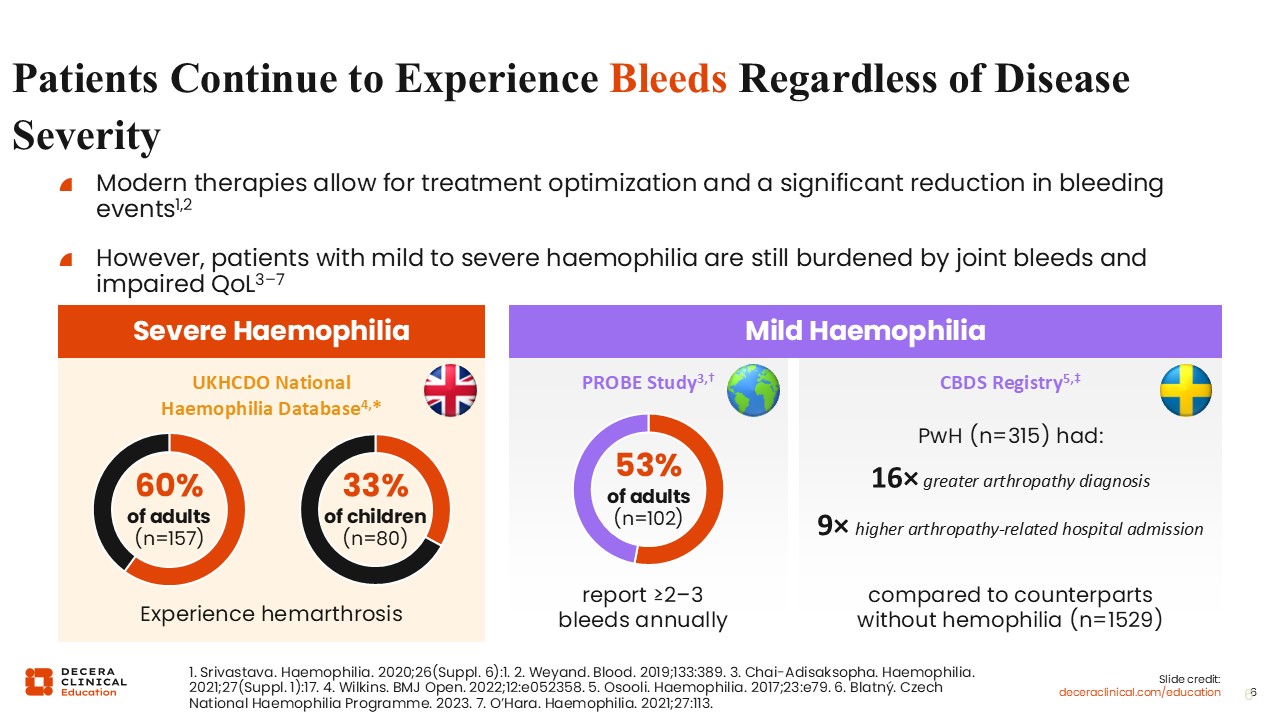
Despite our modern therapies that allow for treatment optimization and a huge reduction in bleeding events compared to prior therapies, patients, regardless of disease severity and regardless of prophylaxis, continue to experience bleeds. This impairs their quality of life.8,9
The UKHCDO National Hemophilia Database from the UK found that in patients with severe hemophilia, 60% of adults and a third of children still experience hemarthrosis.3
This extends to a lesser severity of hemophilia in patients with mild hemophilia. The probe study found 53% of adults reported at least 2-3 bleeds annually, which is clearly much higher than we would aim for.10 The CBDS registry also found that patients with hemophilia were 16 times more likely to have arthropathy and 9 times more likely to have hospital admission related to arthropathy compared to their counterparts without hemophilia.11 Thus, there is room for improvement to avoid bleeds and provide a similar quality of life to those without hemophilia.
Joint Damage Can Occur Even in the Absence of Joint Bleeds
Angela Weyand, MD:
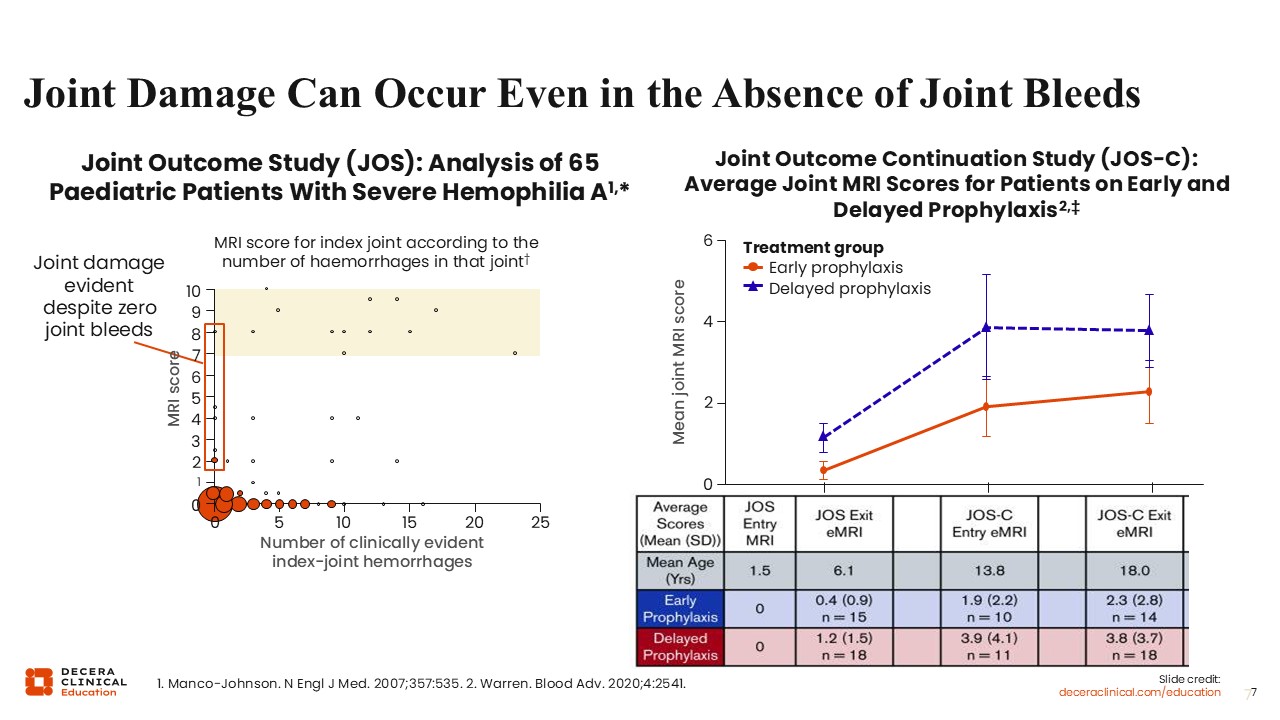
Unfortunately, we know that even in the absence of clinically evident joint bleeding, joint damage is occurring. The joint outcome study was an analysis of 65 pediatric patients with severe hemophilia A. The figure on the left shows the number of clinically evident index joint hemorrhages.12 These are bleeds that the patients identified. The MRI score is here, too. Unfortunately, as shown on the far left of the figure, even patients who had zero clinically evident joint hemorrhages showed elevated scores. The scores show they have joint damage that is occurring, despite being aware of clinically evident bleeding. The figure on the right shows that early prophylaxis is key to preventing this joint damage and that the mean joint MRI score is higher in those with delayed prophylaxis, but does increase even in those started on prophylaxis early.13
Rationale for Targeting Hemostatic Balance to Improve Outcomes in Patients With Hemophilia
Angela Weyand, MD:
As hemophilia therapies have continued to evolve, we have started to target the hemostatic balance to improve outcomes in patients with hemophilia. The rationale for this includes the impact of hemophilia on the maintenance of hemostasis. Procoagulant entities, such as factor VIII and factor IX, which are deficient in hemophilia, are counterbalanced by natural anticoagulants. By targeting these natural anticoagulants, the aim is to restore the hemostatic balance. 14
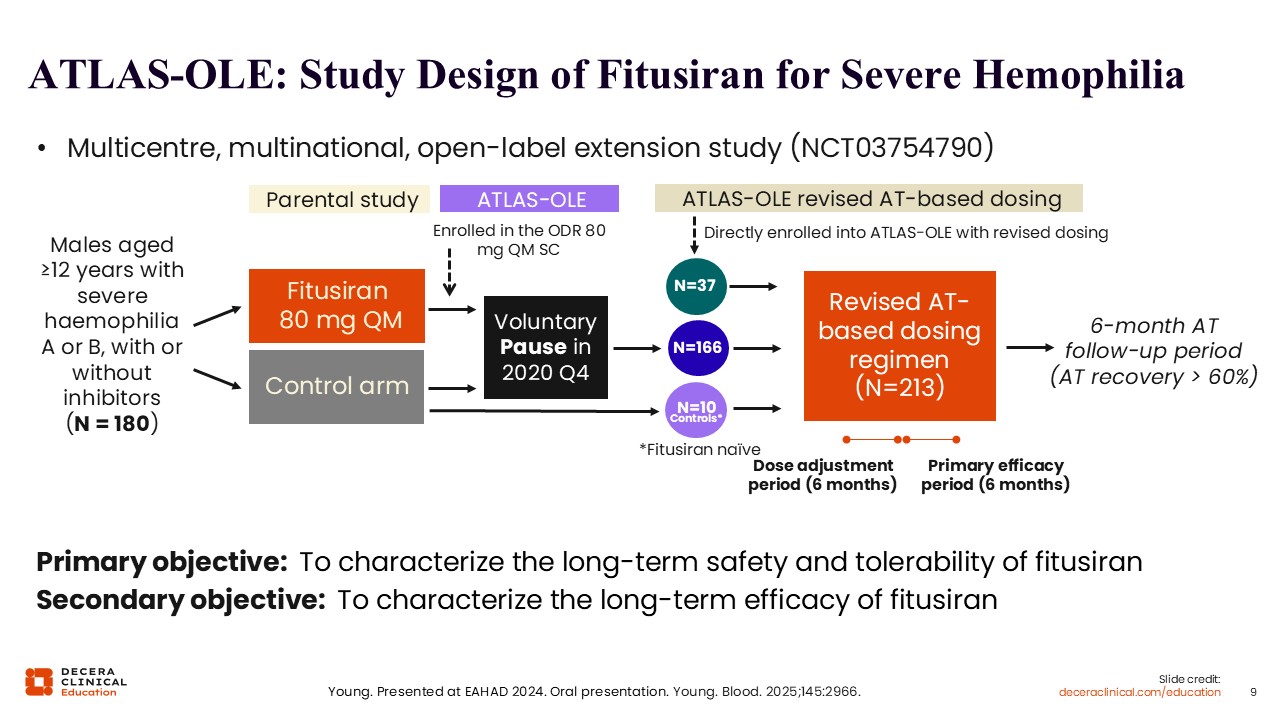
ATLAS-OLE: Study Design of Fitusiran for Severe Hemophilia
Guy Young, MD:
Now, let’s discuss the available therapies that target the natural anticoagulants to restore balance. Fitusiran received its FDA approval based on results from the Atlas OLE, or open-label extension study. The purpose of the open-label extension study was to characterize the long-term safety and efficacy of fitusiran. Essentially, this study is comprised of a group of patients who all came in from prior phase III studies, when they were using the original dose regimen (ODR), which was 80 mg of fitusiran subcutaneously once per month. However, there were thrombotic events with the prior phase III studies. Therefore, a new dosing schema was started after a voluntary pause.15
Atlas-OLE included most of the patients from the parent study as well as those who started the new dosing regimen, which was based on antithrombin levels. Rather than a flat dose, patients would get a flat dose of 50 mg once subcutaneously every 2 months, but then the dose would be adjusted based on the antithrombin levels.16 The point of doing that was to minimize the risk for thrombosis because in the parent studies, the patients who had thrombotic events all had antithrombin levels around 10%.
The new dosing schema led to the lower end of the therapeutic range being antithrombin levels of 15% to minimize the risk for thrombosis and the upper end being 35% because above that range, there is no effect on reducing bleeding. 15
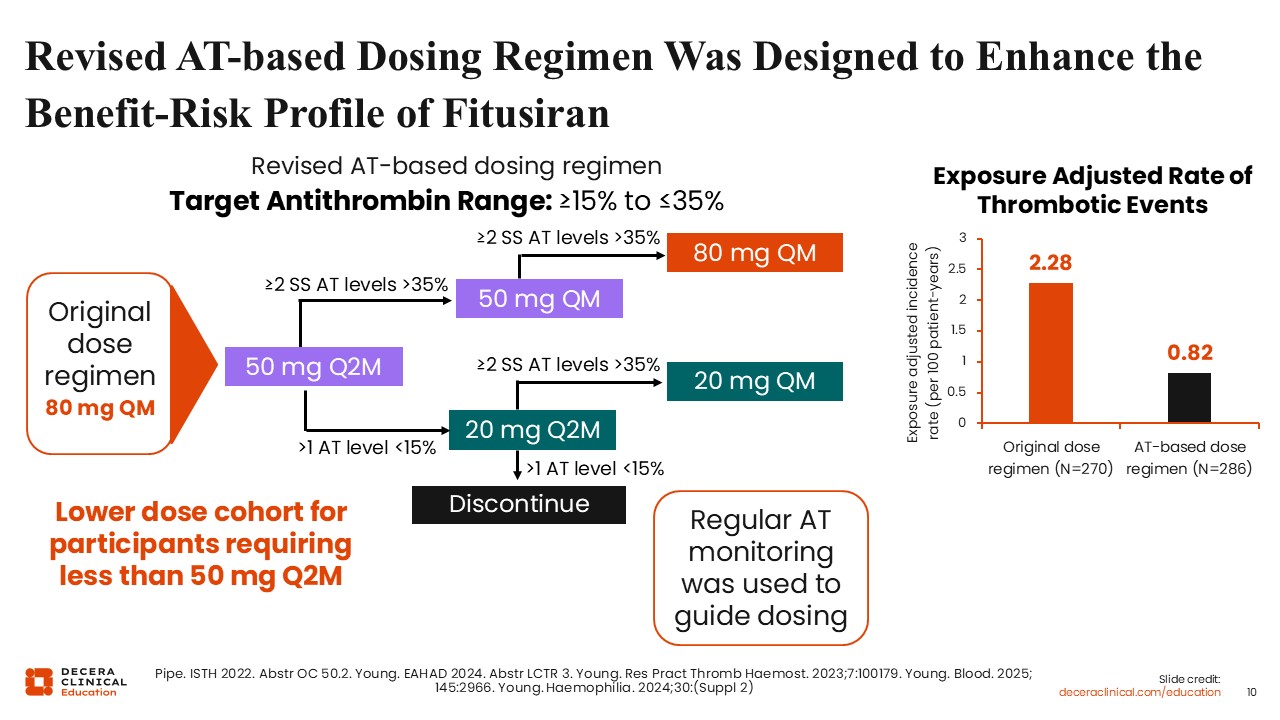
Revised AT-Based Dosing Regimen Was Designed to Enhance the Benefit–Risk Profile of Fitusiran
Guy Young, MD:
Depicted here is the design of the open-label extension study dosing regimen. The new dosing regimen is called the antithrombin-based dosing regimen (ATDR). In the schema depicted here, the original dose regimen was 80 mg monthly. At the top, the ATDR targets an antithrombin range between 15% and 35%. The dosing schema starts all the patients at 50 mg every 2 months and then adjusts the levels, if needed, to increase the frequency to 50 mg every month if the antithrombin level remains above 35% or to reduce the dosing to 20 mg every 2 months if the antithrombin level is below 15%.17
After that, if the antithrombin level is still below 15%, the patients would be discontinued from the study. In the prescribing information, patients may go down to a dose of 10 mg.
If patients at 20 mg every 2 months have an antithrombin level greater than 35%, the dose can be adjusted to 20 mg every month. With this scheme, regular monthly monitoring of antithrombin levels is done.
Did this new dosing regimen improve the safety of the study? The answer is yes. In the ODR, the thrombotic events are measured as per 100 patient-years, and the exposure-adjusted incidence rate was 2.28. For patients on the new ATDR, the rate was 0.82.15,17
In addition to the reduction in thrombosis, there were other adverse events with the ODR, which included gallbladder issues, cholecystitis, and cholelithiasis, as well as increases in the alanine aminotransferase (ALT) and aspartate aminotransferase (AST).15 The new dosing regimen also substantially reduced the rate of cholecystitis and cholelithiasis, and also substantially reduced the rate of ALT and AST elevations compared to the ODR.
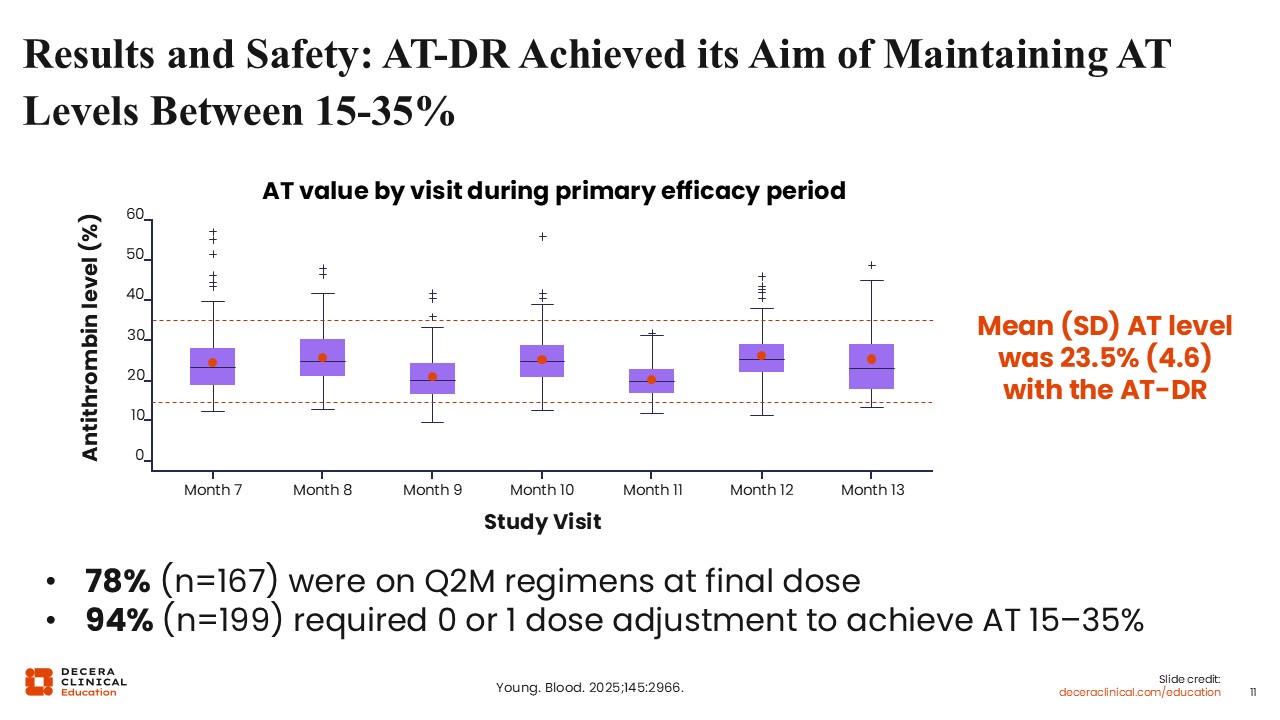
The AT-DR Achieved Its Aim of Maintaining AT Levels Between 15% and 35%
Guy Young, MD:
Did this new ATDR dosing regimen work? In other words, did it achieve the goal of maintaining antithrombin levels between 15% and 35%? The answer is yes. Starting at Month 7, but certainly by Month 13, most were between 15% and 35%, with a mean antithrombin level of 23.5%. Most of the patients were able to remain on an every-2-month regimen. Nearly all patients needed no adjustments or just 1 dose adjustment. 15
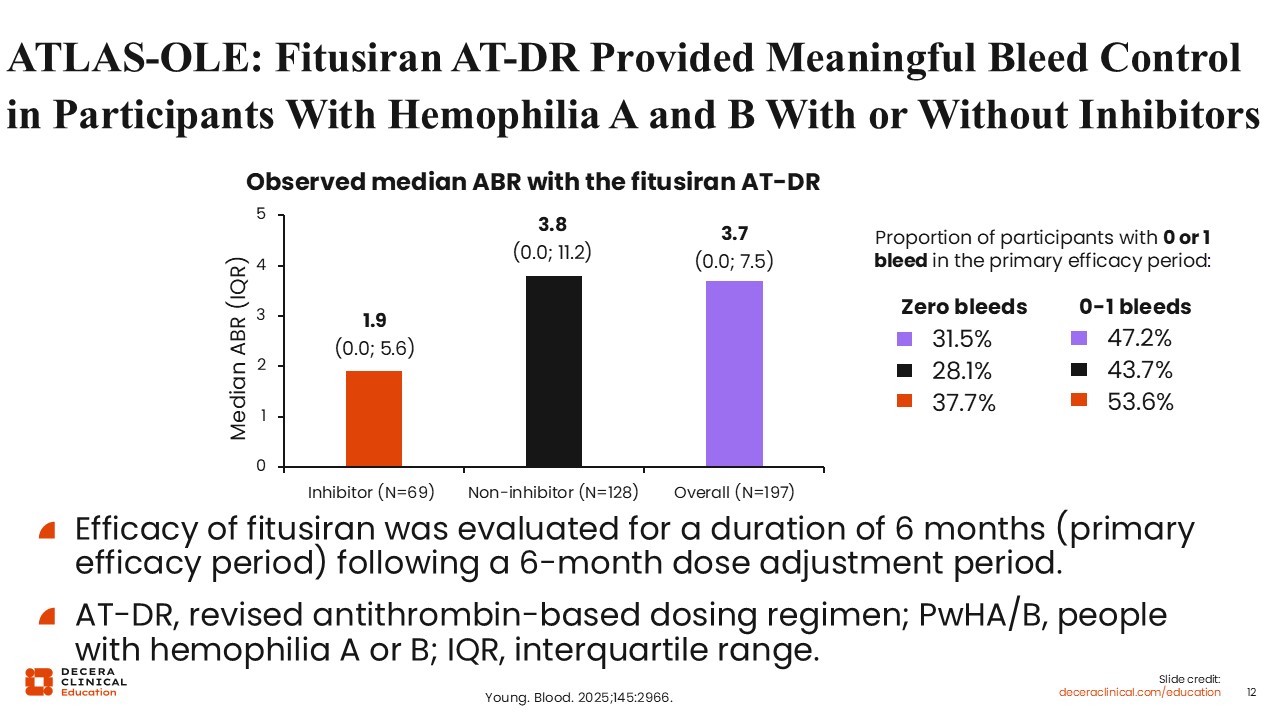
ATLAS-OLE: Fitusiran AT-DR Provided Meaningful Bleed Control in Participants With Hemophilia A and B With or Without Inhibitors
Guy Young, MD:
With respect to efficacy, there was a sacrifice in efficacy for the sake of safety. In the ODR, the median annualized bleeding rate (ABR) was zero.
In Atlas-OLE, the median ABR overall is 3.7. It is 1.9 in patients with inhibitors and 3.8 in patients without inhibitors. However, this was still meaningfully effective bleed protection. On the right shows there were zero bleeds in approximately one third of the patients in the study, and zero or 1 bleed in approximately half the patients.15 In summary, although the new dosing regimen was not as effective as the ODR, it substantially improved the safety while maintaining efficacy considered important and meaningful for patients with hemophilia.
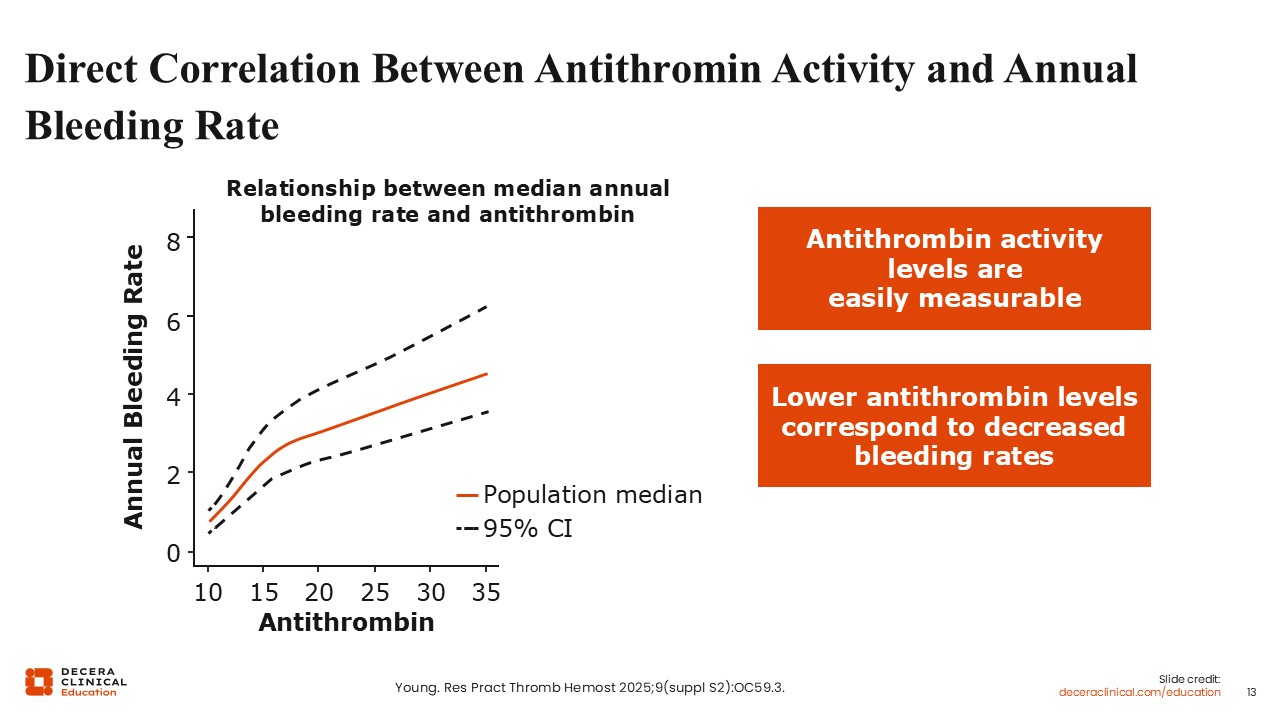
Direct Correlation Between Antithrombin Activity and Annual Bleeding Rate
Guy Young, MD:
There is a direct correlation between antithrombin levels, shown on the X axis, and the ABR on the Y axis. The lower the antithrombin level is driven, the lower the ABR. This comes into play when you're treating patients clinically. For example, if you have a patient who is in the range but having some bleeds, it could be possible to adjust the dose. With a patient who has antithrombin levels at 30%, but their ABR might range from 2-3, you would want to improve that by increasing the dose or increasing the frequency to target a lower ABR.15
Concizumab
Angela Weyand, MD:
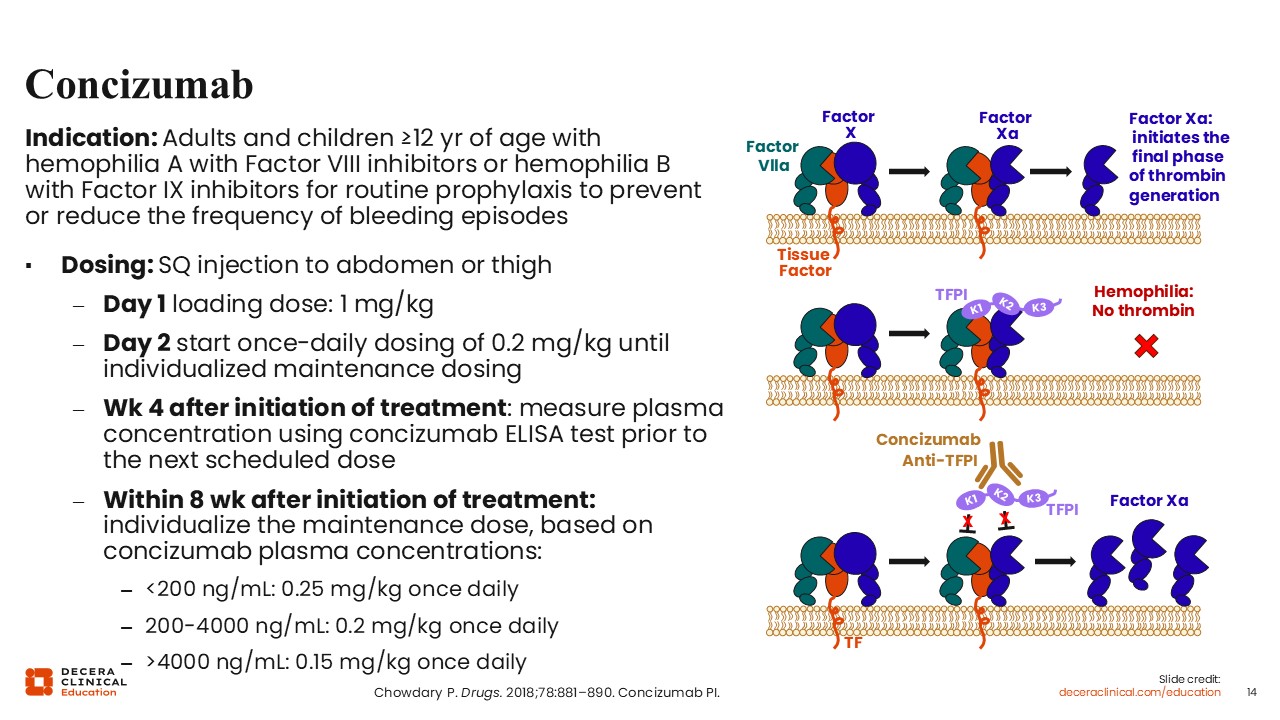
Concizumab is a new rebalancing agent. It is currently indicated for the treatment of adults and children at least 12 years of age with hemophilia A with factor VIII inhibitors or hemophilia B with factor IX inhibitors for routine prophylaxis to prevent or reduce the frequency of bleeding. It is an anti-TFPI antibody and its mechanism of action is targeting and binding the Kunitz-2 (K2) domain of TFPI, a natural anticoagulant, which prevents it from interacting with factor. This enhances the generation of thrombin. Concizumab is dosed subcutaneously and via injection to the abdomen or leg and starts with a loading dose on Day 1, followed by once daily dosing. At Week 4 after initiation of treatment, plasma concentrations are measured prior to the next scheduled dose to individualize the maintenance dose for patients based on that concizumab plasma concentration.18
Concizumab Phase III Trials: Efficacy at 56-Wk Cutoff (Arms 1-4)
Angela Weyand, MD:
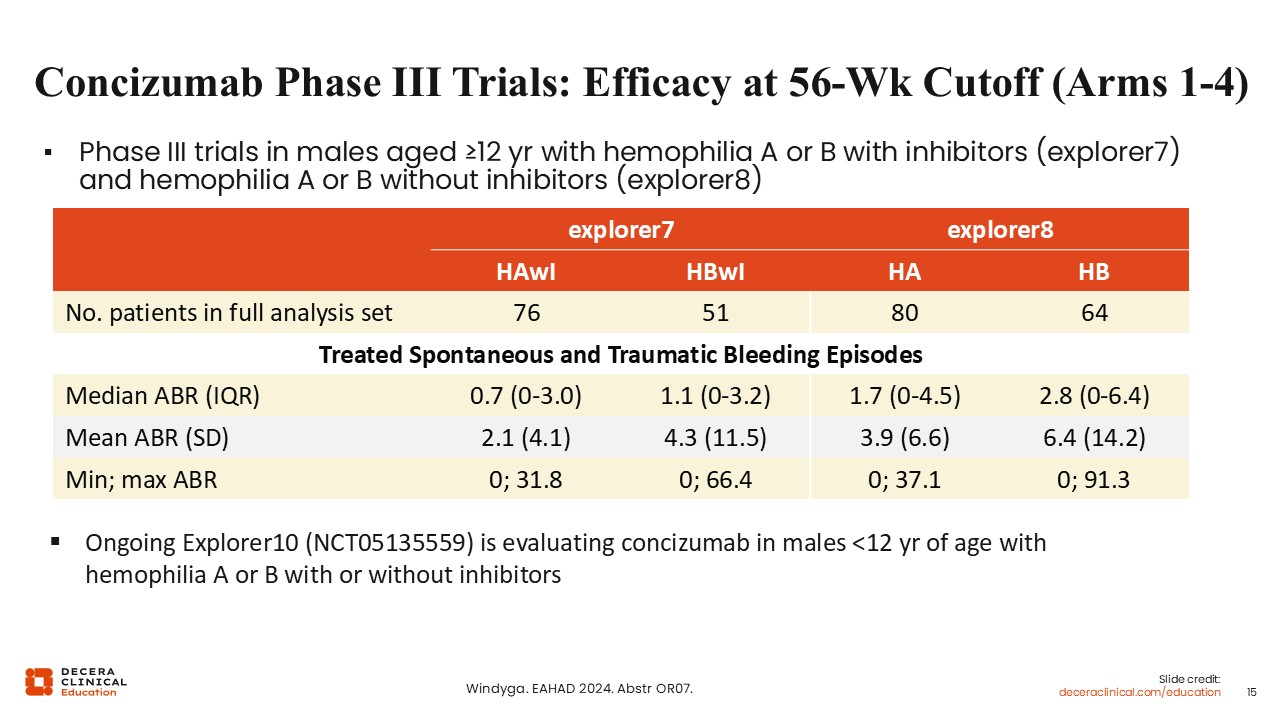
The concizumab phase III trials looked at efficacy at a 56-week cutoff in males with hemophilia A or B with inhibitors and individuals at least 12 years of age with hemophilia A or B without inhibitors. explorer7 enrolled patients with inhibitors, and explorer8 enrolled patients without inhibitors. The chart shows the treated spontaneous and traumatic bleeding episodes. The median ABR for hemophilia A with inhibitors was 0.7, hemophilia B with inhibitors 1.1, and 1.7 or 2.8 without inhibitors for hemophilia A and B, respectively. In the 2 cohorts without inhibitors, there are ongoing studies evaluating concizumab in males less than 12 years of age with hemophilia A or B without inhibitors.19
Concizumab Phase III Trials: Safety at 56-Wk Cutoff (Arms 1-4)
Angela Weyand, MD:
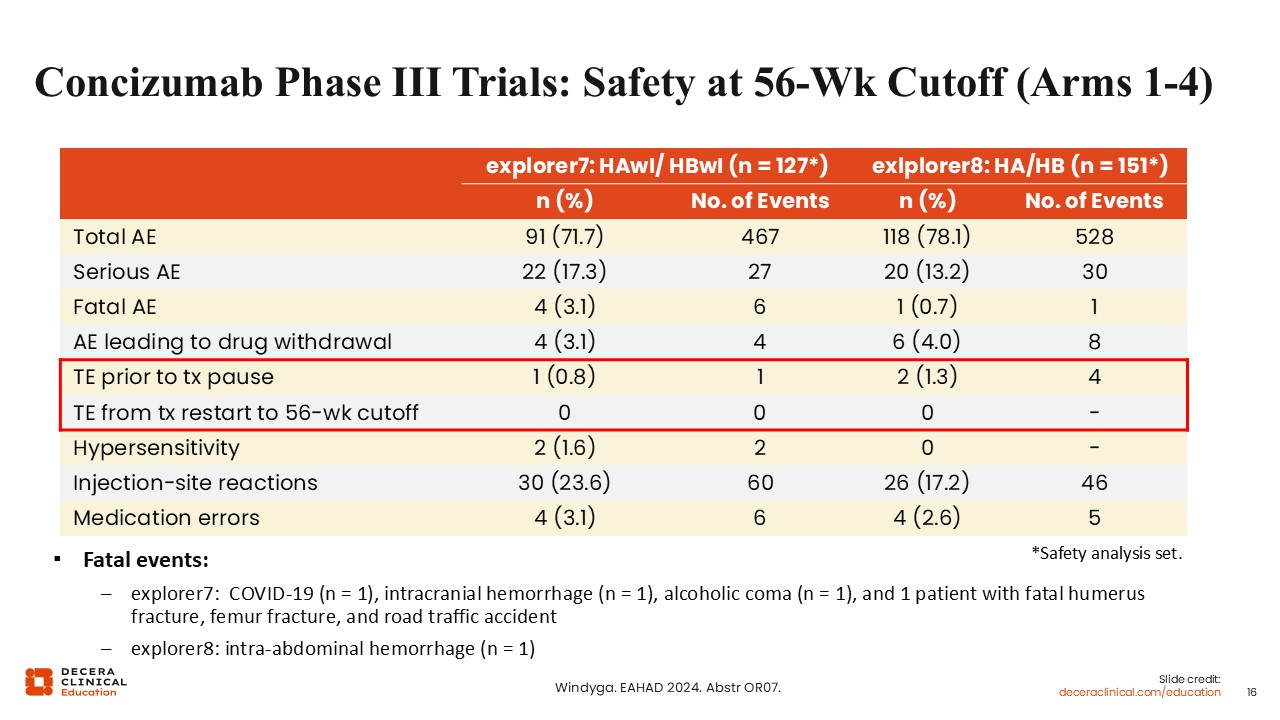
The safety of concizumab was evaluated at the 56-week cut-off in both the inhibitor arms of explorer7 as well as the noninhibitor arms of explorer8. Overall, most of the adverse events were observed in similar trials. Thromboembolic events occurred prior to a treatment pause, with a few in each cohort, but following treatment restart to the 56-week cutoff, there were no further events of this type. There were some fatal events within explorer7. There was 1 patient with an intracranial hemorrhage, and others related to COVID infection, an alcoholic coma, and a traffic accident. In explorer8, there was 1 fatal event of intra-abdominal hemorrhage.19
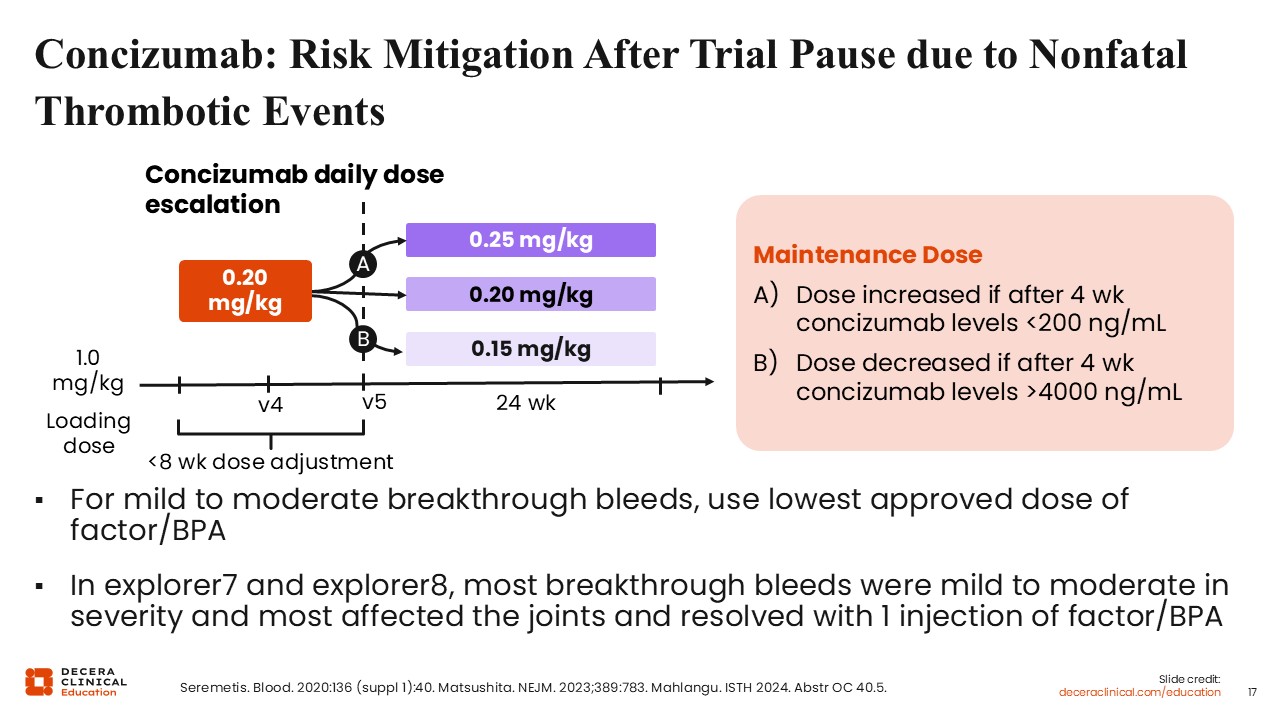
Concizumab: Risk Mitigation After Trial Pause due to Nonfatal Thrombotic Events
Angela Weyand, MD:
Thromboembolic events that occurred initially in the explorer7 and explorer8 trials caused a trial pause so that risk mitigation could occur due to these nonfatal thrombotic events. Following the pause, a risk mitigation strategy was created and is outlined here.20 To mitigate risk, there is a loading dose, followed by a dose adjustment based on the concizumab level drawn at the 4-week mark after treatment initiation.21 Based on that concizumab level, if the level was low, the dose would be increased. If it was higher than 4000 ng/mL, the dose would be decreased, and if it was between 200-4000 ng/mL, it would remain the same. There were further recommendations for mild to moderate breakthrough bleeds, including using only the lowest approved dose of factor or a bypassing agent for treatment of those bleeds.22 Of note from the explorer trials, most of the breakthrough bleeds were mild to moderate in severity, so that was not an issue and most affected joints that had bleeds resolved with 1 injection of factor or a bypassing agent.
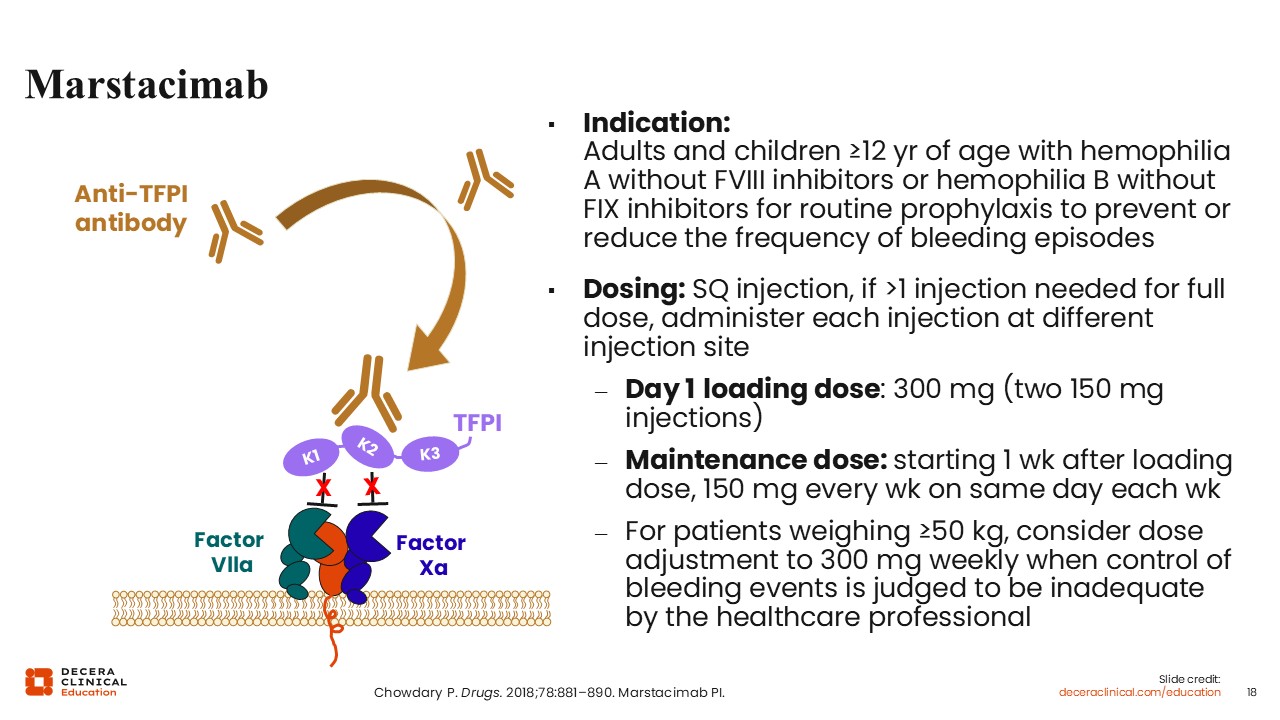
Marstacimab
Angela Weyand, MD:
Marstacimab is another anti-TFPI antagonist antibody that is directed against the K2. Marstacimab is indicated for routine prophylaxis to prevent or reduce the frequency of bleeding episodes in adult and pediatric patients 12 years of age or older with hemophilia A without factor VIII inhibitors or hemophilia B without factor IX inhibitors. Similar to other antibody treatments, it is given via subcutaneous injection. If more than 1 injection is needed, the injections are recommended to be given at different injection sites. Dosing starts off with a Day 1 loading dose, which is fixed at 300 mg and administered as 2 separate 150 mg injections. Then, 1 week after the loading dose, a maintenance dose of 150 mg follows every week by subcutaneous injection on the same day each week. For patients weighing more than 50 kg, it is recommended to consider dose adjustments to 300 mg weekly if the control or prevention of bleeding episodes is deemed to be inadequate by the healthcare professionals.23
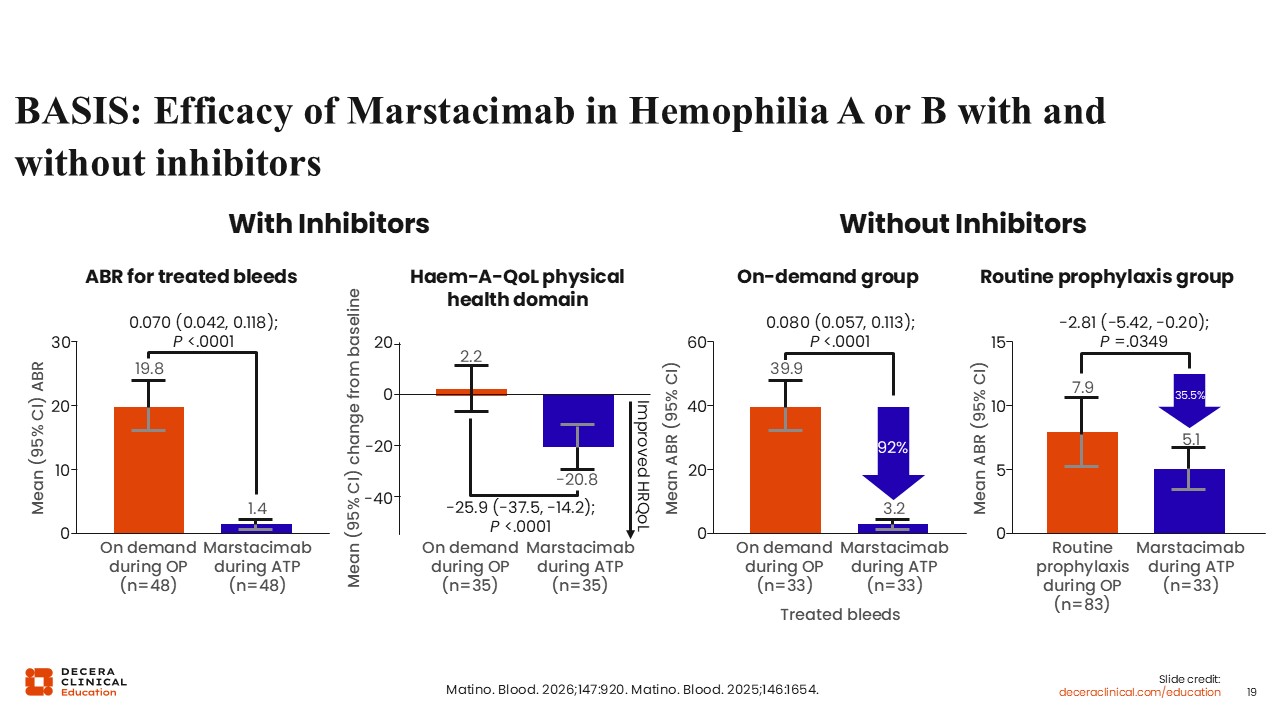
BASIS: Efficacy of Marstacimab in Hemophilia A or B With and Without Inhibitors
Angela Weyand, MD:
The BASIS clinical trials looked at the efficacy and safety of marstacimab in hemophilia A or B with and without inhibitors. Shown here are the ABRs for treated bleeds in the patient population with inhibitors. Initially, patients using on-demand therapies came in with high ABRs, almost 20, yet once they started on the active treatment phase with marstacimab, the ABR decreased to 1.4.
Concurrently with the ABR improvement, there was a significant improvement in the “Haem-A-QoL” physical health domain, which looks at quality of life related to physical health in patients with hemophilia and those without inhibitors.24,25
In the on-demand group, patients who were not previously receiving prophylaxis came in with very high ABRs of almost 40, which decreased substantially (92%) with prophylaxis and marstacimab. Patients who had previously been on routine prophylaxis with other factor products came in with a high ABR of 7.9, which decreased to 5.1 during the active treatment phase with marstacimab.24,25
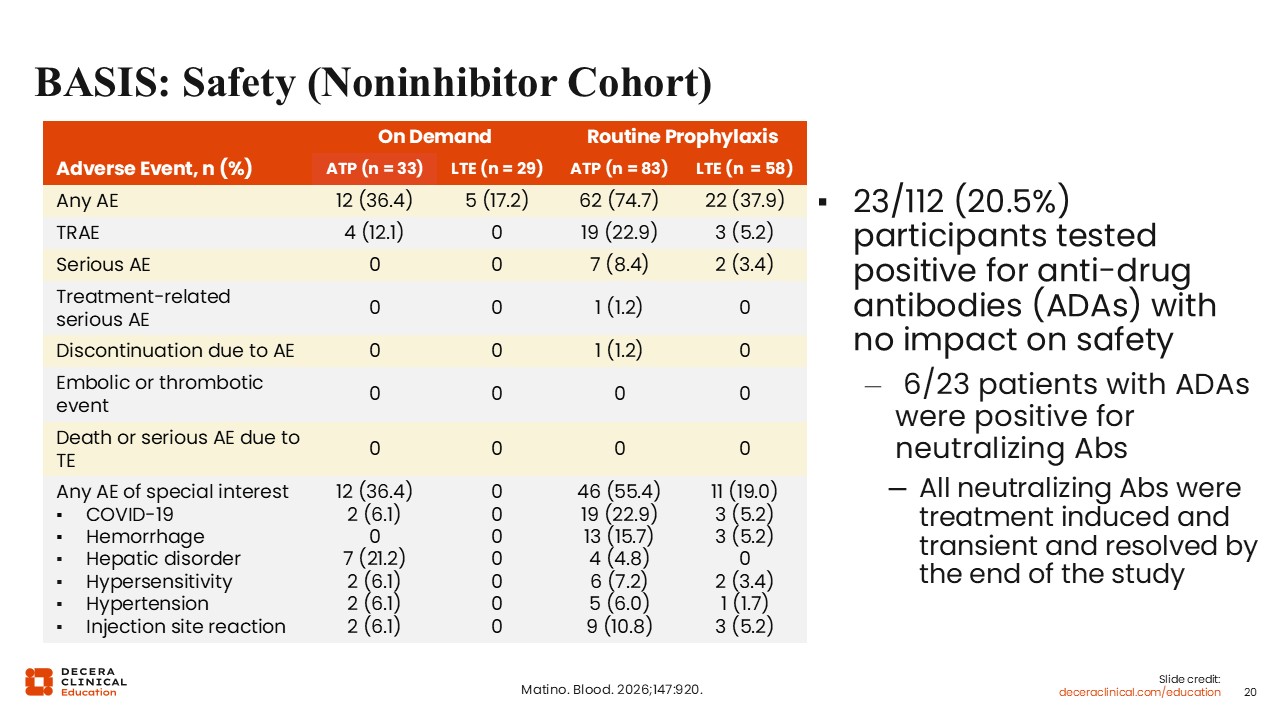
BASIS: Safety (Noninhibitor Cohort)
Angela Weyand, MD:
In addition, the BASIS trial looked at safety. This table outlines the safety events in the noninhibitor cohort, which were similar to observations from other studies of prophylaxis in patients with hemophilia. Of note, there were no embolic or thrombotic events within the noninhibitor cohort. Of interest, approximately one fifth of participants tested positive for antidrug antibodies to marstacimab, but these had no impact on safety.24
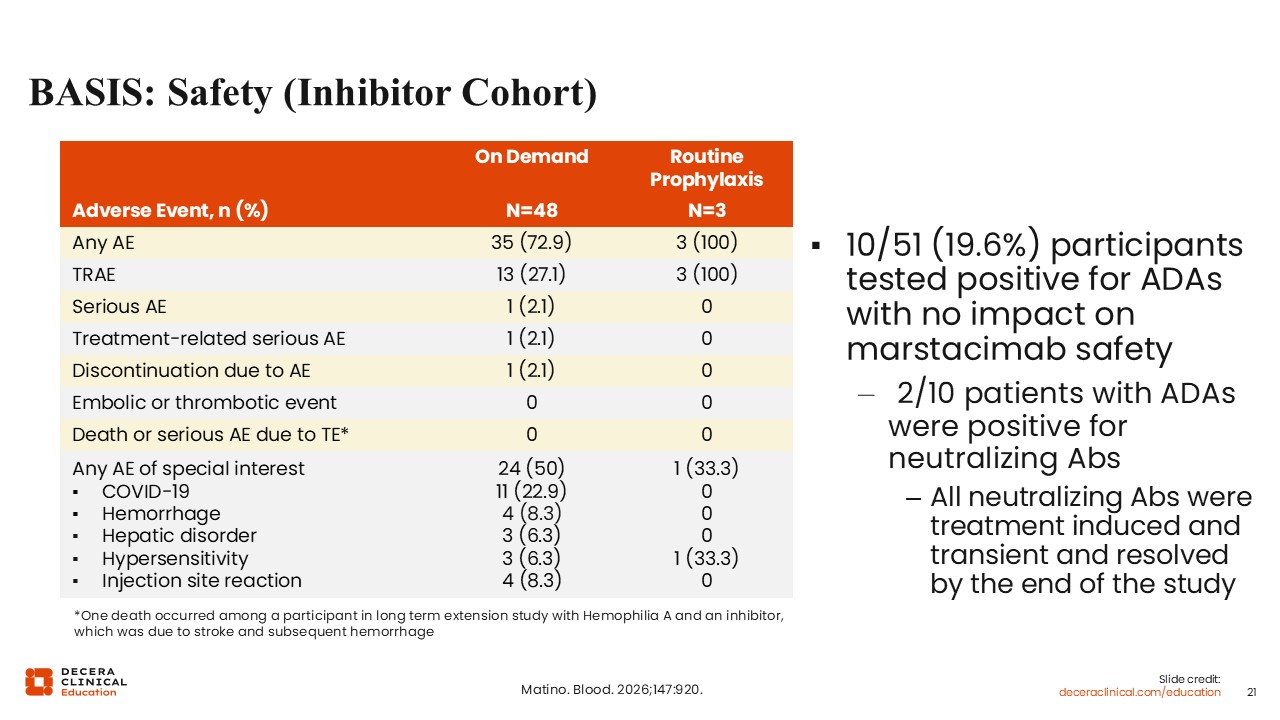
BASIS: Safety (Inhibitor Cohort)
Angela Weyand, MD:
The inhibitor cohort safety data from the BASIS trial are outlined here. There were no embolic or thrombotic events. There were no deaths or serious adverse events due to thromboembolism. There was 1 treatment-related serious adverse event.
In the adverse events of special interest, there were a few reports of hemorrhage, hepatic disorder, hypersensitivity, and injection-site reaction. Of note again in the inhibitor cohort, like the noninhibitor cohort, approximately one fifth of participants tested positive for antidrug antibodies, but there was no impact on the safety of marstacimab.24
Properties of Rebalancing Agents
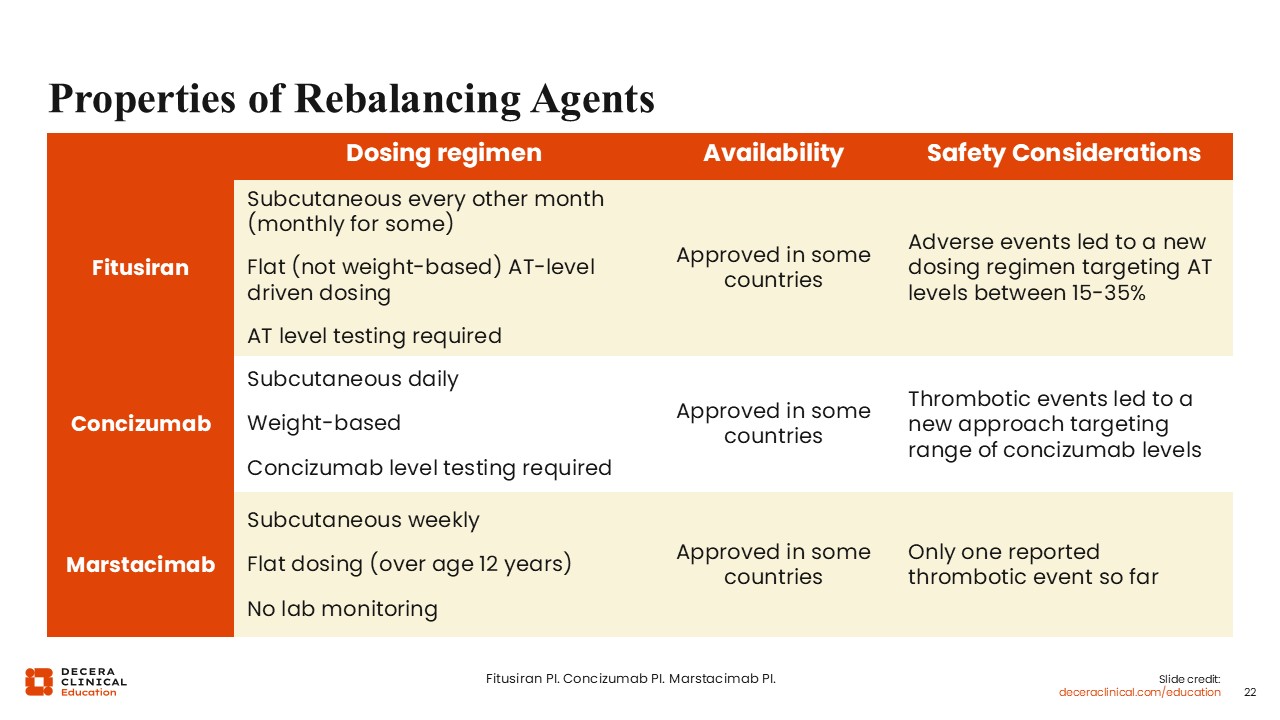
Guy Young, MD:
This slide shows a comparison of the 3 recently approved rebalancing agents, fitusiran, concizumab, and marstacimab. Fitusiran is given subcutaneously every 2 months, although some patients would have to increase to every month.16 Concizumab is given subcutaneously once per day.18 Marstacimab is given subcutaneously once per week.23 The other aspect is the dosing. Marstacimab has flat dosing, meaning it is not based on the patient's weight. However, it is adjusted based on the antithrombin levels, but marstacimab has no required laboratory monitoring.
Concizumab is weight based, with each patient having their own specific dose based on their weight. Concizumab requires lab testing 1-4 weeks after the first dose, and this would result in potentially adjusting the dose of concizumab based on the levels again. Patients with the highest levels had a higher risk for thrombosis, so this was a risk mitigation strategy.18
For fitusiran, antithrombin level testing is required, which means that it requires laboratory monitoring once per month for the first 6 months and then annually thereafter. The monitoring is necessary to adjust the dose to maintain the antithrombin level between 15% and 35%.16
These medications are approved in the United States for similar hemophilia indications, although marstacimab is not yet approved for patients with inhibitors. Otherwise, these 3 drugs are approved for patients 12 years of age or older with hemophilia A or B with and without inhibitors, except for marstacimab, which is not yet approved for patients with inhibitors, although that will likely change in the future.
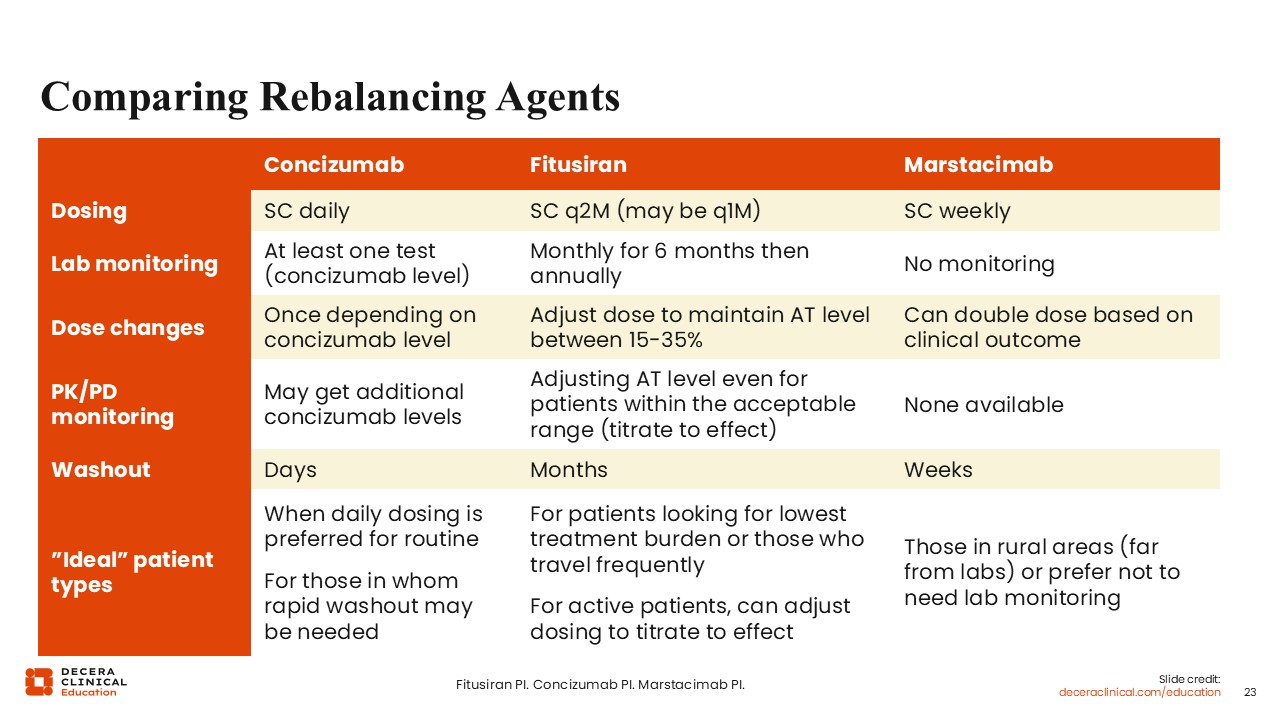
Comparing Rebalancing Agents
Guy Young, MD:
In contrast to concizumab and fitusiran, marstacimab has no required laboratory monitoring, but there is an option to double the dose for patients who are having at least 2 bleeds, in approximately 6 months. One caveat is that patients must weigh 50 kg to be eligible to double the dose. Marstacimab is approved for patients 12 years of age or older, but they also must weigh at least 35 kg.
For pharmacokinetic or pharmacodynamic monitoring with concizumab and fitusiran, monitoring is required. Patients can get additional dosing beyond what is written in the label, based on need. For marstacimab, there is no lab test available. Any clinical decision regarding whether to continue or discontinue marstacimab, or to double or not double the dose, is not based on laboratory tests.
For the washout, concizumab has the shortest half-life, and its washout is measured in days. Marstacimab takes several weeks to wash out, whereas fitusiran takes 4-6 months. This is important to note in patients for whom a rapid washout might be needed, such as those who may require multiple surgeries in the near future. In that case, concizumab may be ideal. In other cases, fitusiran may be indicated for a patient who is looking for the least intensive dosing regimen or a patient who travels a lot because it's only dosed every 2 months. Because there's no lab monitoring required, patients who don't like to have lab testing or who live far away from a lab might be the ones for whom marstacimab would be an ideal fit.16,18,23
There are some patients who present with special considerations that are weighed heavily to guide treatment. For example, let’s say a 14-year-old with severe hemophilia B started on an extended half-life factor IX concentrate. Now he starts getting busier in his life and wants an easier administration method while maintaining at least equal efficacy for bleed control. To reduce his treatment burden, a healthcare professional might discuss fitusiran or marstacimab as options with the patient and his parents, also depending on inhibitors.
In another example, A 36-year-old with severe hemophilia A with inhibitors currently on emicizumab started a new job that requires him to travel 80% of the time. It is challenging for him to carry the vials of emicizumab along with injection supplies, especially since emicizumab must be refrigerated. He is seeking an option that will be the best to accommodate his travel schedule. This is another occasion where, depending on inhibitors, fitusiran or marstacimab may be a worthwhile discussion.
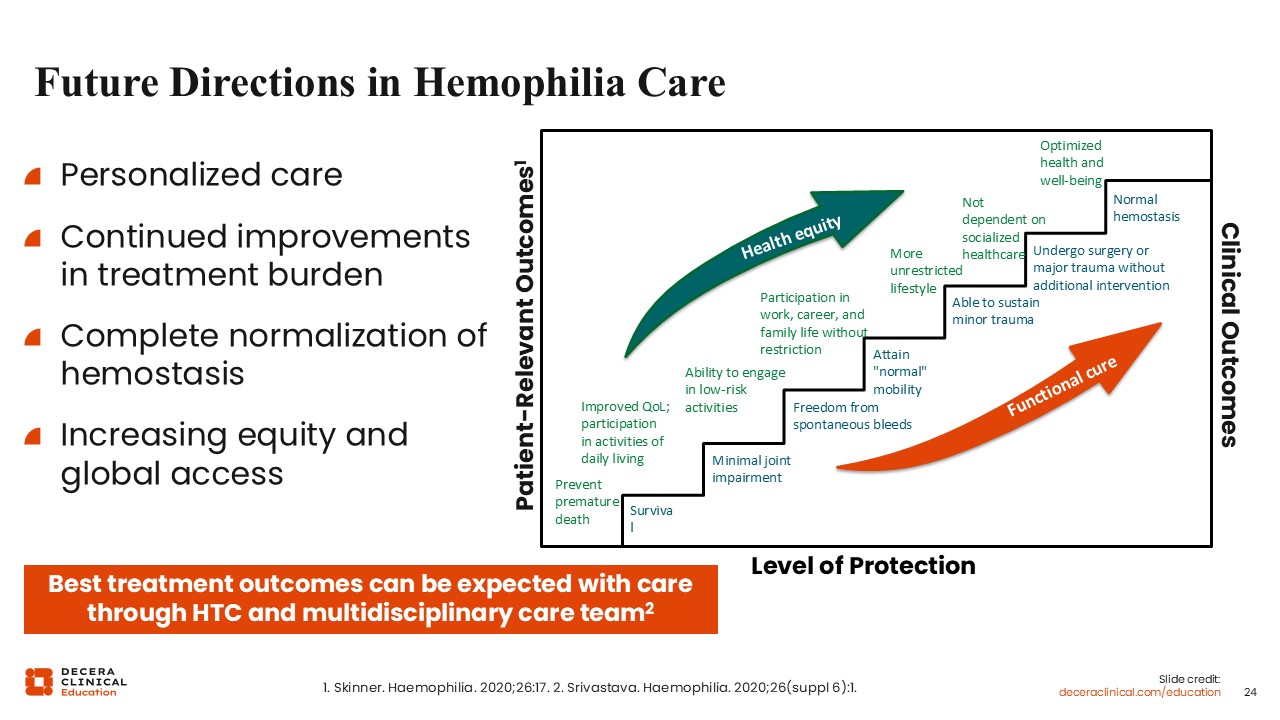
Future Directions in Hemophilia Care
Guy Young, MD:
For future directions, we are stepping towards health equity in patients with hemophilia and working towards a functional cure. The clinical field has moved far along this curve and is at the level of patients having a more unrestricted lifestyle in terms of health equity. Patients are not dependent on socialized healthcare and are moving towards optimized health and well-being.
With current therapies, clinical medicine is closer to achieving normal hemostasis among patients with hemophilia. At this point, we are probably able to have patients who sustain minor trauma and undergo minor surgery or major trauma without additional intervention. For major surgeries, a patient with hemophilia will probably still need factor replacement. Therefore, we still have a little way to go, but we are certainly moving far along with some of these new medications.