CE / CME
Expert Analysis: Clinical Advances Journal Club on Emerging TROP-2 and TIGIT Therapies in Lung Cancer
Physicians: Maximum of 1.00 AMA PRA Category 1 Credit™
European Learners: 1.00 EBAC® CE Credit
Released: January 22, 2026
Expiration: July 21, 2026
Activity
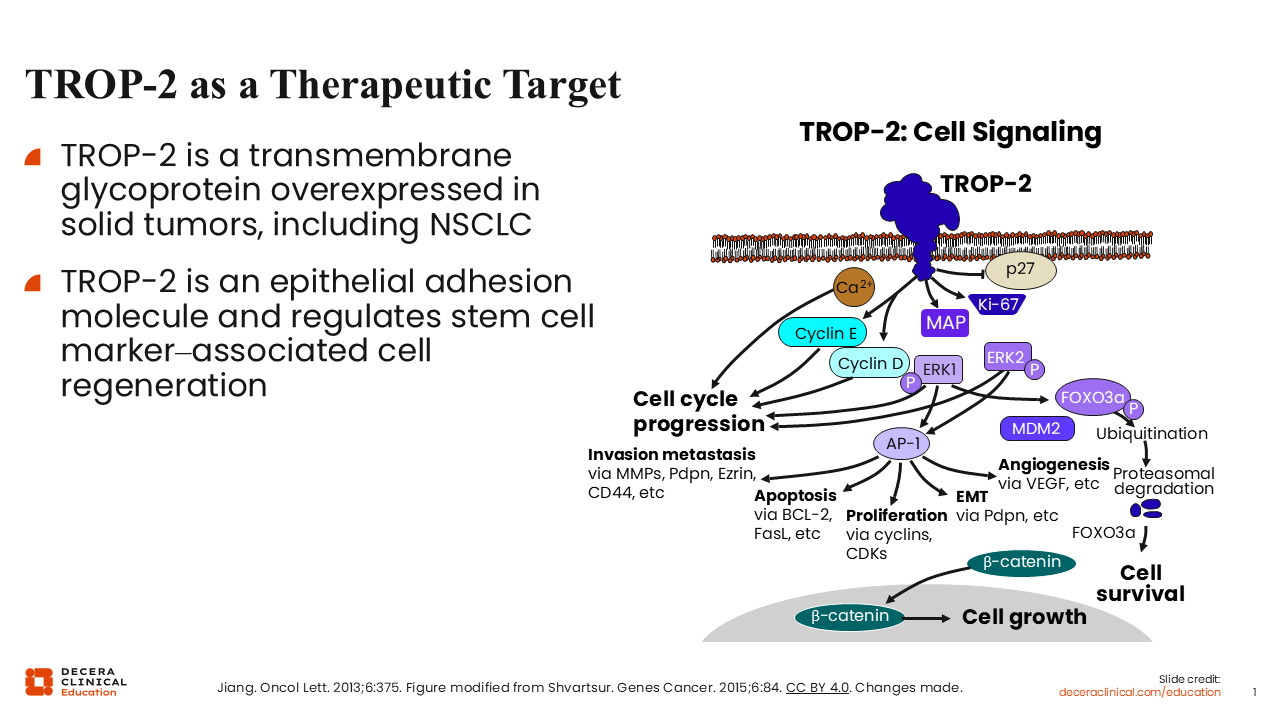
TROP-2 as a Therapeutic Agent
Benjamin Levy, MD, FASCO:
TROP-2 is emerging as a potential therapeutic target as we know that TROP-2 is expressed ubiquitously on solid tumors including lung cancer.1,2 TROP-2 overexpression is also associated with worse outcomes in NSCLC.1 Fortunately, we now have several therapies in development that target this protein.
Anti–TROP-2 ADCs
Benjamin Levy, MD, FASCO:
The first of the TROP-2–targeting agents was the ADC SG, followed by Dato-DXd and, more recently, sac-TMT.3-5 All of these ADCs contain a TROP-2 IgG1 antibody with high-affinity binding and different topoisomerase-1 inhibitor payloads. Their drug-to-antibody ratios vary between 4.0 and 7.6, and they have different dosing schedules, but we believe they function in a similar way.
TROPION-Lung01 and EVOKE-01 Failed to Show OS Benefit in Previously Treated Advanced or Metastatic NSCLC
Benjamin Levy, MD, FASCO:
The first phase III studies of Dato-DXd and SG, TROPION-Lung01 and EVOKE-01, compared these TROP-2 ADCs to docetaxel in the second-line setting for advanced NSCLC in patient populations that were not enriched for TROP-2 expression.6-8 Unfortunately, neither of these studies showed a survival advantage with these compounds vs the standard of care. Median OS for SG vs docetaxel was 11.3 vs 9.6 months (HR: 0.89; P = .1028).8 Median OS for Dato-DXd vs docetaxel was 12.9 vs 11.8 months (HR: 0.93; P = .530).6 There was a possible PFS benefit for Dato-DXd in the nonsquamous population (5.5 vs 3.6 months; HR: 0.63), but the 2 studies were negative for OS overall. However, further study of these agents has demonstrated promising use for NSCLC despite these disappointing results.
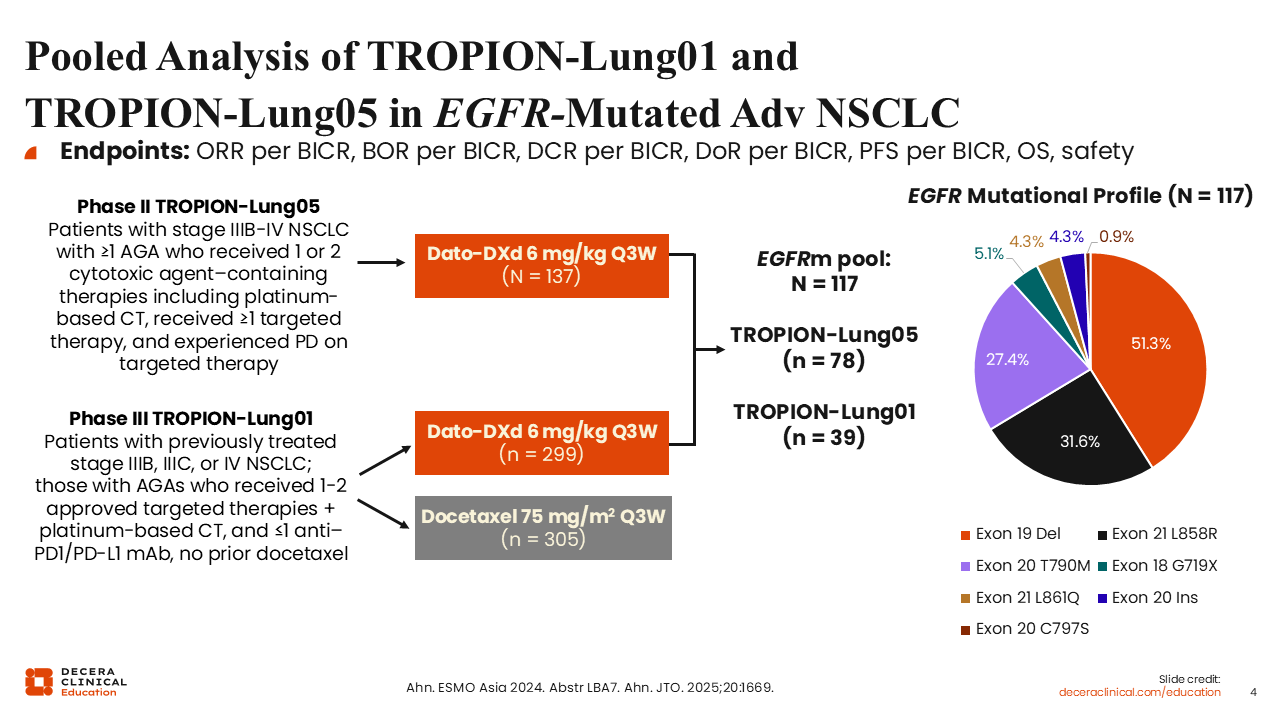
Pooled Analysis of TROPION-Lung01 and TROPION-Lung05 in EGFR-Mutated Adv NSCLC
Benjamin Levy, MD, FASCO:
A pooled analysis of the TROPION-Lung01 and the single-arm phase II TROPION-Lung05 examined the efficacy of Dato-DXd in 117 patients with EGFR-mutated advanced NSCLC.9,10 The vast majority of pooled patients had the EGFR mutations exon 19 del, exon 21 L858R, or exon 20 T790M.
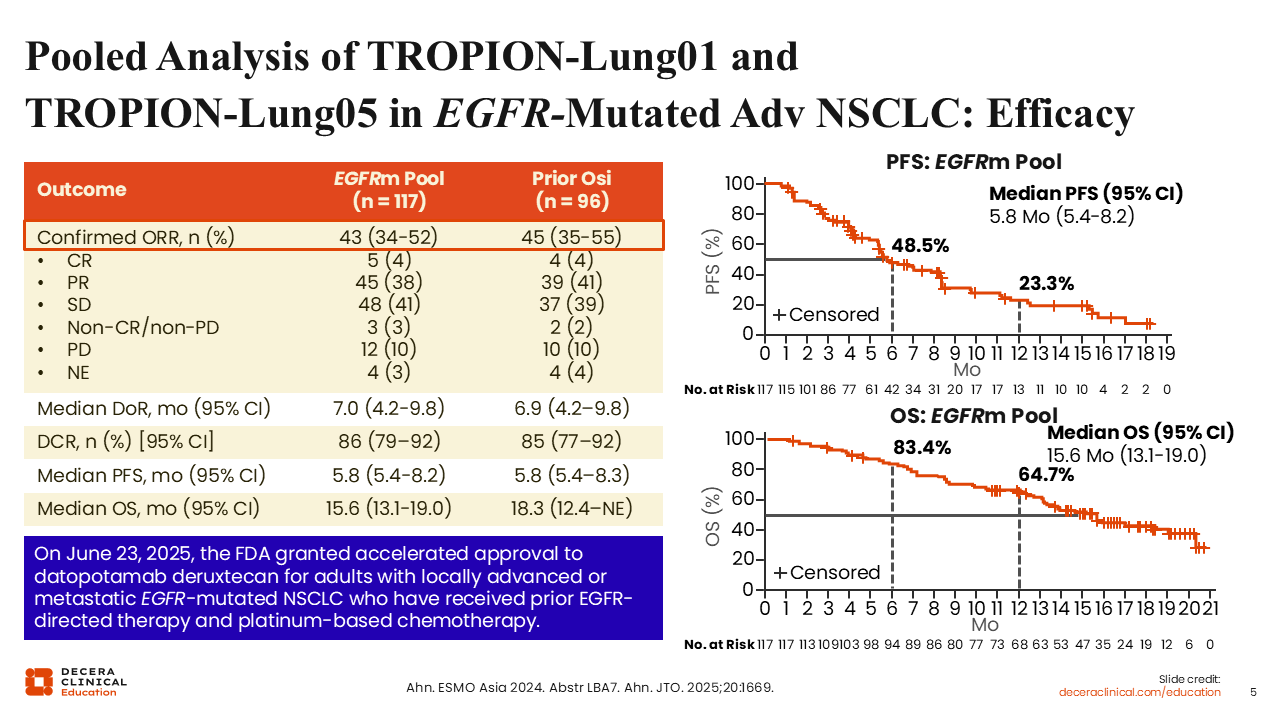
Pooled Analysis of TROPION-Lung01 and TROPION-Lung05 in EGFR-Mutated Adv NSCLC: Efficacy
Benjamin Levy, MD, FASCO:
Overall response rate (ORR) was 43% and 45% in patients with prior osimertinib treatment, which were encouraging results in a pretreated group of patients that are EGFR positive.9,10 In the prior osimertinib population, median duration of response, PFS, and OS in the total population were 6.9, 5.8, and 18.3 months, respectively. These results led to FDA approval of Dato-DXd for adults with locally advanced or metastatic EGFR-mutated NSCLC who have received prior EGFR-directed therapy and platinum-based CT on June 23, 2025.11
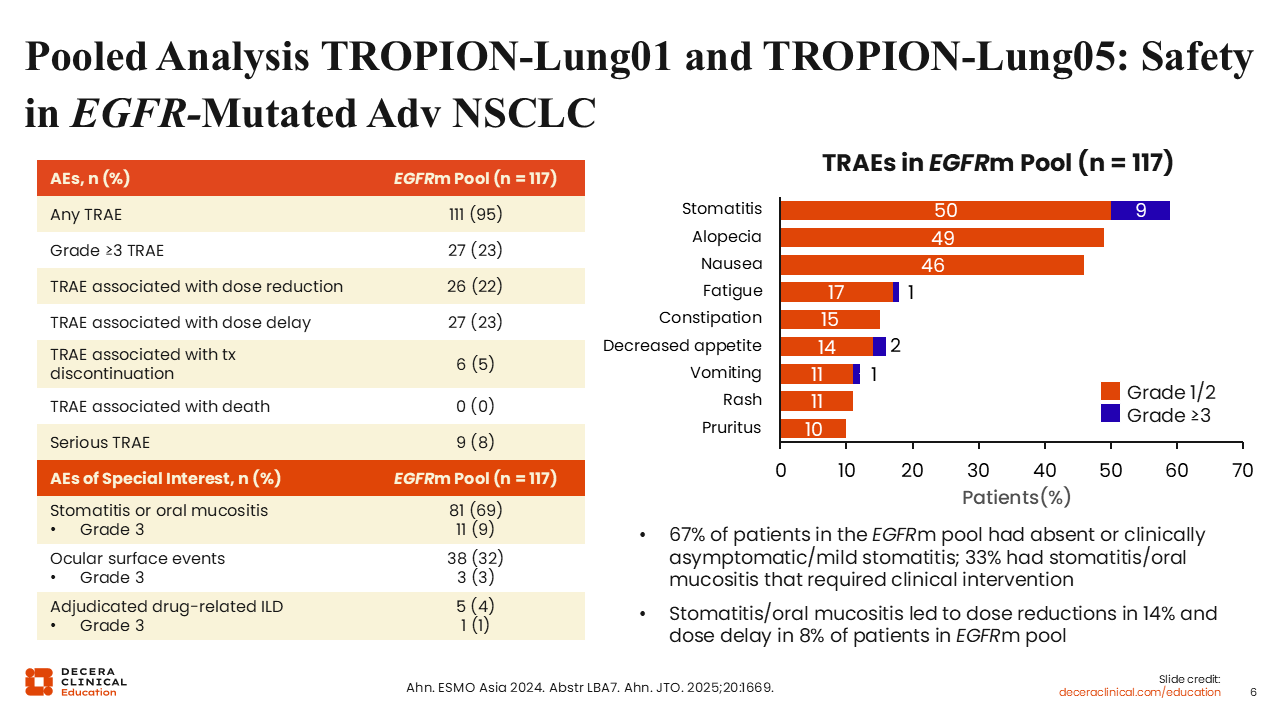
Pooled Analysis of TROPION-Lung01 and TROPION-Lung05: Safety in EGFR-Mutated Adv NSCLC
Benjamin Levy, MD, FASCO:
This analysis did not reveal any new safety signals with Dato-DXd in the EGFR-mutant patient population compared with the general NSCLC populations of these studies.6,9,10,12 Of note, stomatitis, alopecia, and nausea are the most common adverse events (AEs) that we see with Dato-DXd. Other AEs of special interest include oral mucositis, ocular surface events, and drug-related interstitial lung disease/pneumonitis. Interstitial lung disease is inherently observed with all of the TROP-2 ADCs. It is an uncommon event in this disease setting, but it is important to be aware of it.
TROPION-Lung02: Phase I Study of Dato-DXd + Pembro ± Platinum CT in Advanced NSCLC
Benjamin Levy, MD, FASCO:
I had the pleasure of presenting this analysis of the phase Ib TROPION-Lung02 study, which evaluated the safety and tolerability of Dato-DXd in combination regimens in the first line for patients with advanced or metastatic NSCLC.13 Patients did not have actionable genomic alterations (eg, EGFR, ALK, ROS1). Two dose levels of Dato-DXd were combined with pembrolizumab with and without platinum-based CT, carboplatin, and cisplatin in 6 cohorts.
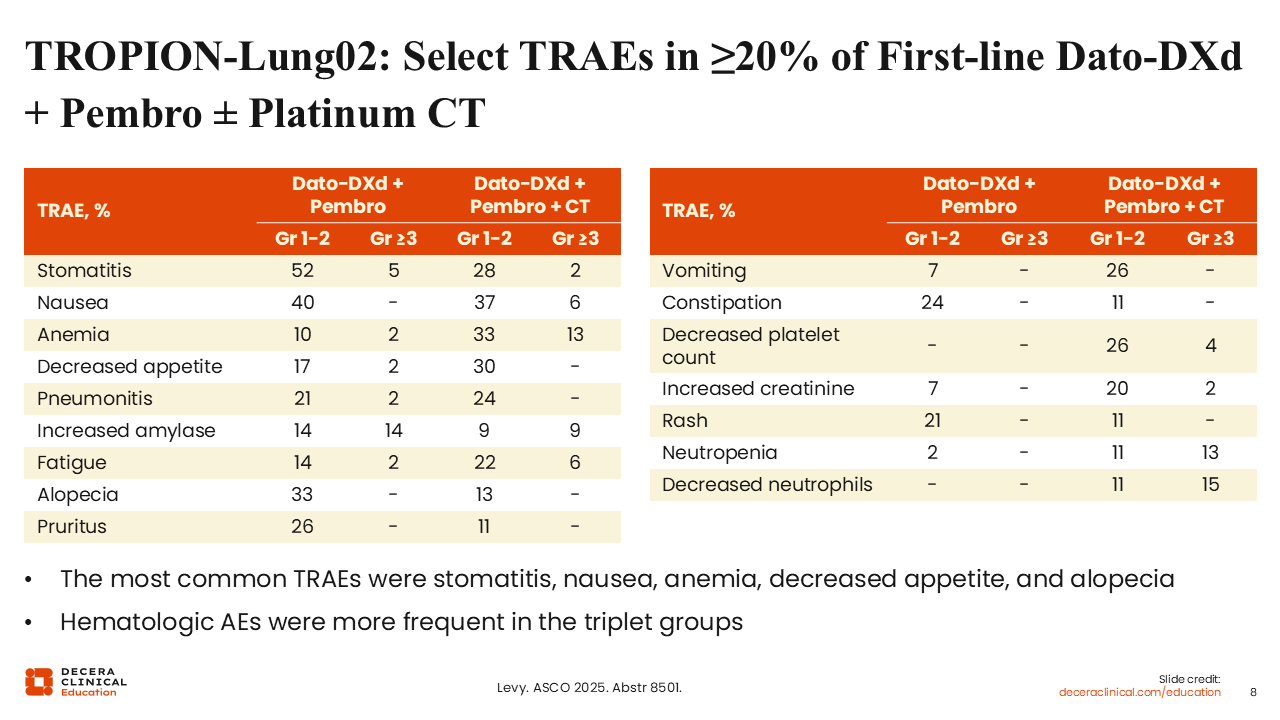
TROPION-Lung02: Select TRAEs in ≥20% of First-line Dato-DXd + Pembro ± Platinum CT
Benjamin Levy, MD, FASCO:
We observed that cytopenias (eg, anemia, decreased platelet count, and neutropenia) were more frequent with Dato-DXd combined with platinum and pembrolizumab compared with Dato-DXd with only pembrolizumab.13 Stomatitis was again one of the most common treatment-related AEs across regimens.
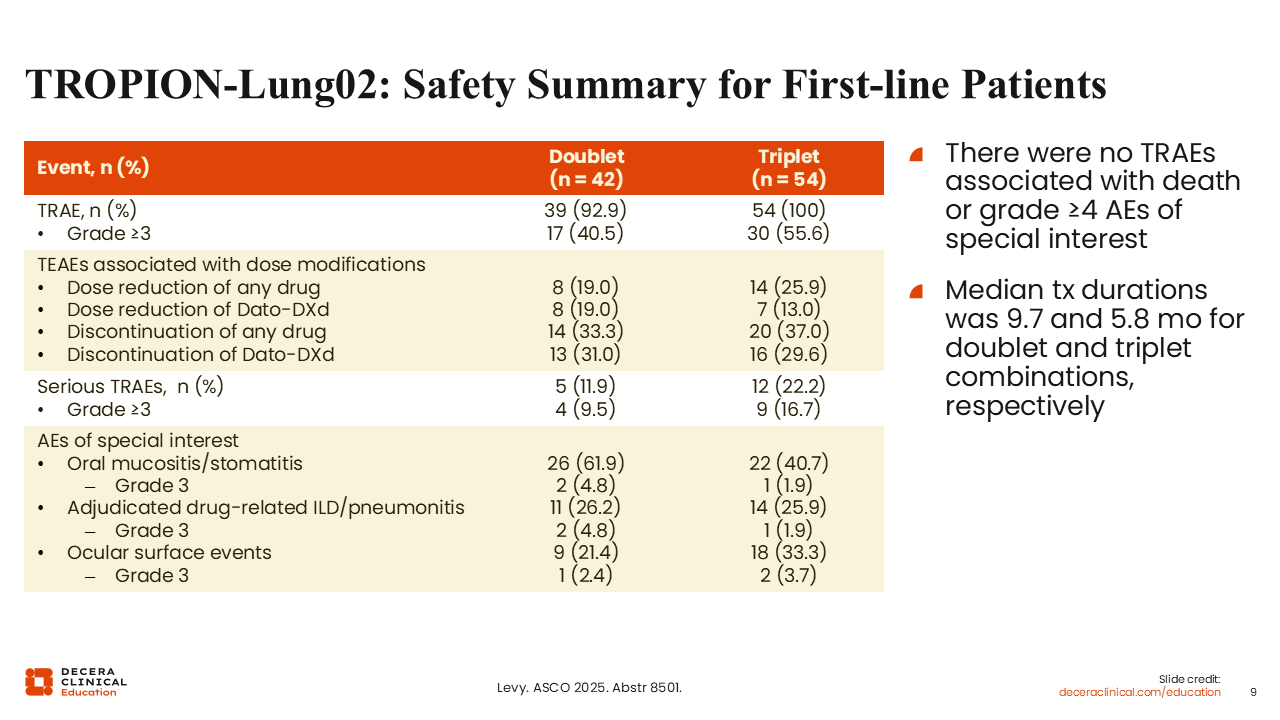
TROPION-Lung02: Safety Summary for First-line Patients
Benjamin Levy, MD, FASCO:
Rates of grade 3 pneumonitis were quite low (4.8% for the doublet regimens and 1.9% for the triplet regimens).13 Overall, the Dato-DXd combinations were reasonably well tolerated in the frontline. I would like to see if these regimens are better tolerated than pemetrexed.
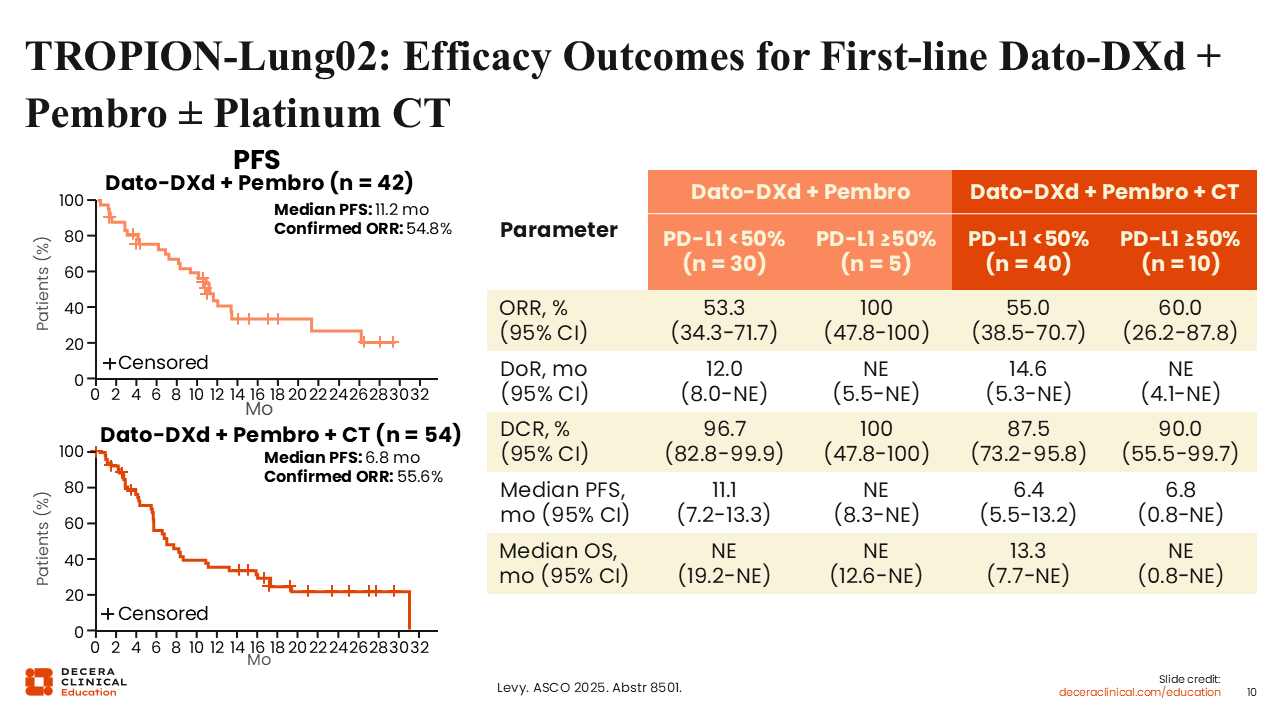
TROPION-Lung02: Efficacy Outcomes for First-line Dato-DXd + Pembro ± Platinum CT
Benjamin Levy, MD, FASCO:
The efficacy outcomes were also encouraging. ORR for the doublet and triplet regimens was 55% and 56%, respectively.13 Median PFS was 11.2 and 6.8 months. Promising results were also observed across treatment arms and PD-L1 expression levels (<50% and ≥50%). There are several ongoing studies aiming to confirm these results in larger data sets.
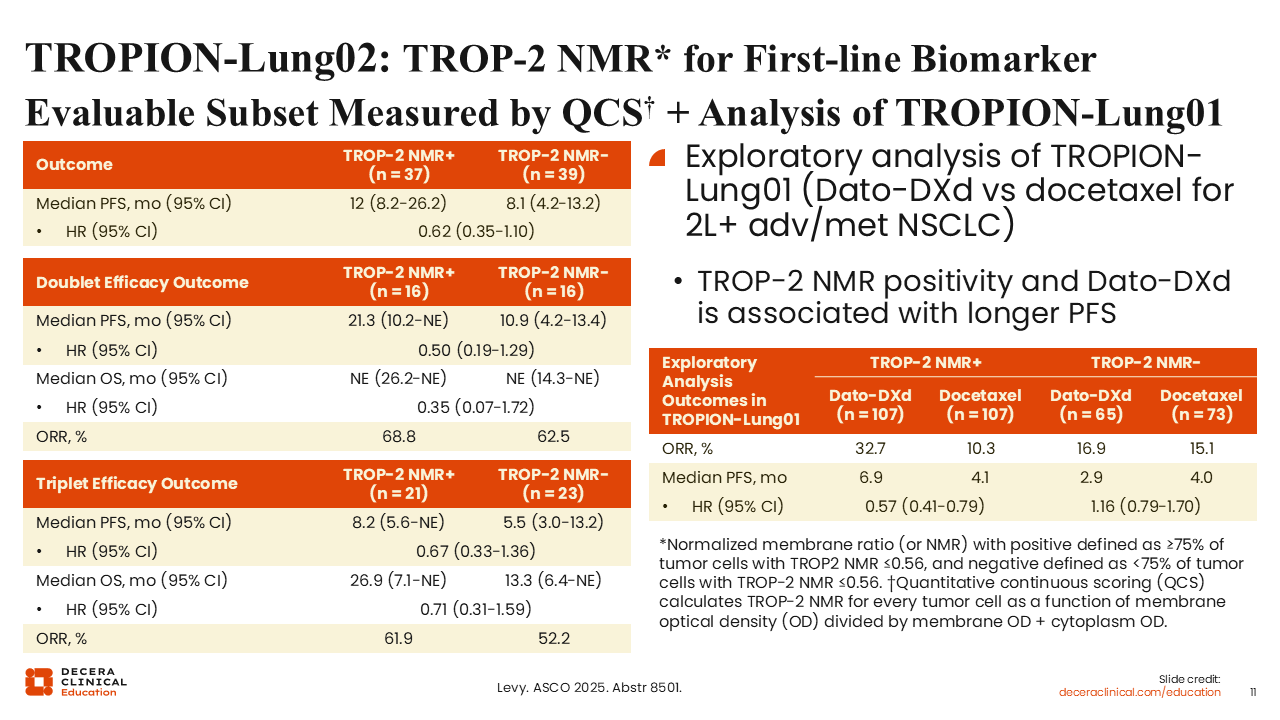
TROPION-Lung02: TROP-2 NMR for First-line Biomarker Evaluable Subset Measured by QCS + Analysis of TROPION-Lung01
Benjamin Levy, MD, FASCO:
This report also included an analysis of TROPION-Lung02 looking into tumor membrane expression of TROP-2.13 We used digital computational pathology to have artificial intelligence read images of histology slides and evaluate TROP‑2 expression on the membrane of tumor cells as well as internally. We hypothesize that these TROP-2 ADCs function best by being internalized, so more TROP-2 expressed internally than externally could predict efficacy.
The computed normalized membrane ratio (NMR) did seem to enrich for better outcomes with Dato-DXd. NMR positive and NMR negative were defined as ≥75% and <75% of tumor cells with TROP-2 NMR ≤0.56, respectively. Median PFS for TROP-2 NMR-positive patients was 12 months compared with 8.1 months for TROP-2 NMR-negative patients (HR: 0.62).
We also performed an exploratory analysis of the TROPION-Lung01 data and found that TROP-2 NMR positivity predicted longer median PFS for Dato-DXd vs TROP-2 NMR negativity (6.9 vs 2.9 months; HR: 0.57). The computational pathology did not predict a difference for the docetaxel arm (TROP-2 NMR positive vs NMR negative: 4.1 vs 4.0 months; HR: 1.16).
This method of biomarker analysis is new, but I believe that eventually something like this approach could be used in clinical practice for TROP-2 ADCs or other agents.
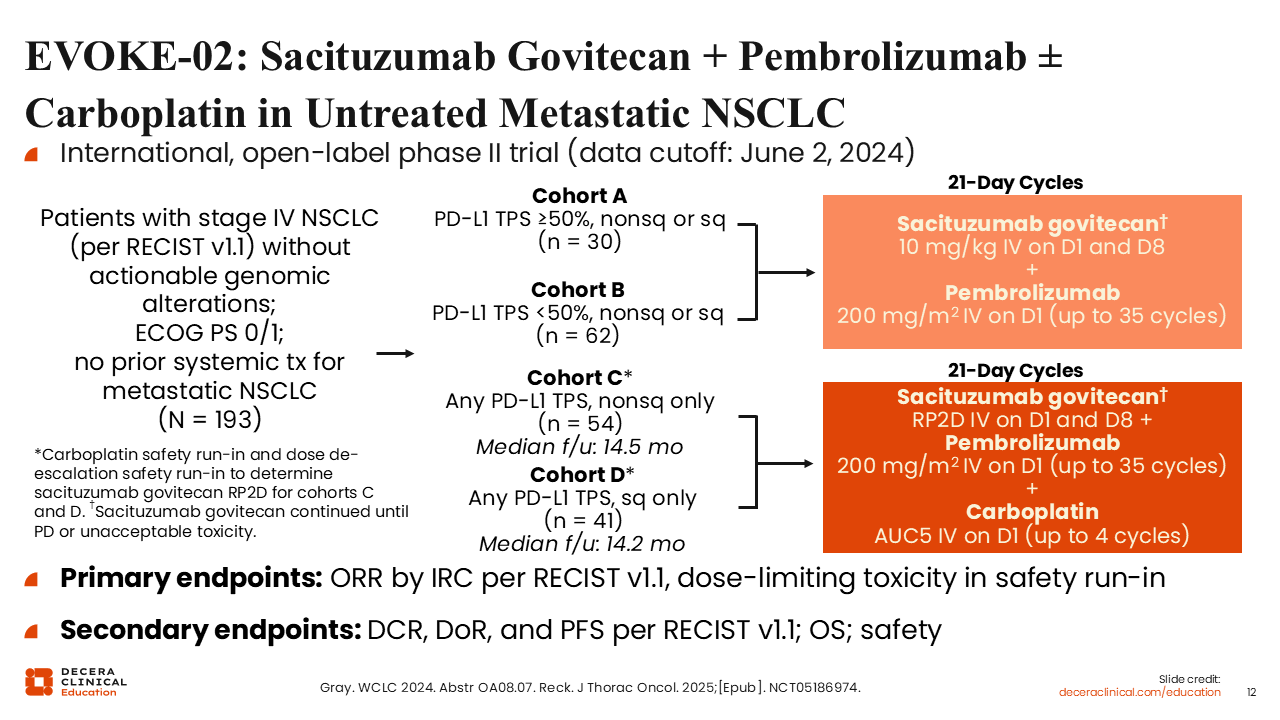
EVOKE-02: SG + Pembrolizumab ± Carboplatin in Untreated Metastatic NSCLC
Benjamin Levy, MD, FASCO:
The phase II EVOKE-02 trial had a very similar design as TROPION-Lung02, in this case using SG.14,15 Cohort A and B evaluated SG plus pembrolizumab, and cohorts C and D looked at SG plus pembrolizumab and platinum. Cohorts A and B enrolled 30 patients with PD-L1 TPS ≥50% and 62 patients with PD-L1 TPS <50%, respectively, whereas cohorts C and D enrolled 54 patients with nonsquamous NSCLC and 41 patient with squamous NSCLC, respectively. Primary endpoints were ORR by independent review committee and dose-limiting toxicity.
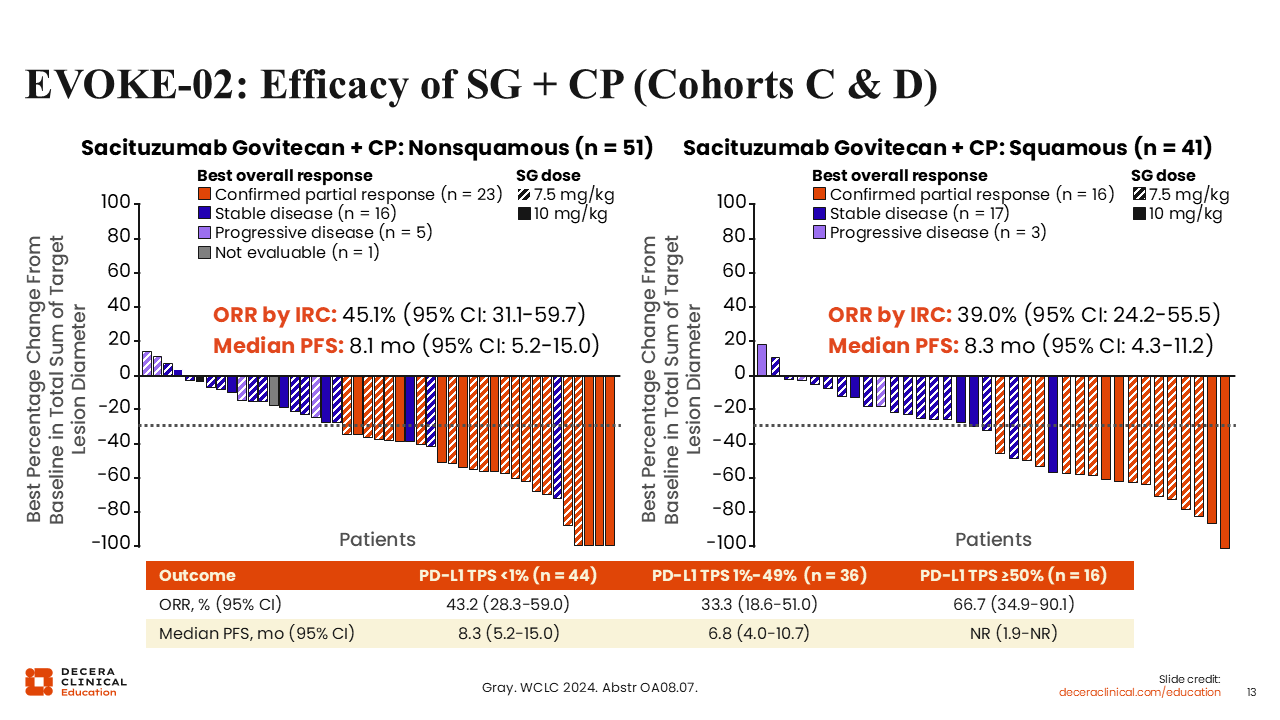
EVOKE-02: Efficacy of SG + CP (Cohorts C & D)
Benjamin Levy, MD, FASCO:
Cohorts C and D saw similar efficacy outcomes in the nonsquamous and squamous subpopulations (ORR: 45.1% and 39.0%; median PFS: 8.1 and 8.3 months).14 There were also encouraging response rates across PD-L1 levels. We will need further study to determine if these combinations can outperform standard-of-care treatments like pemetrexed for adenocarcinoma or taxanes for squamous cell carcinoma.
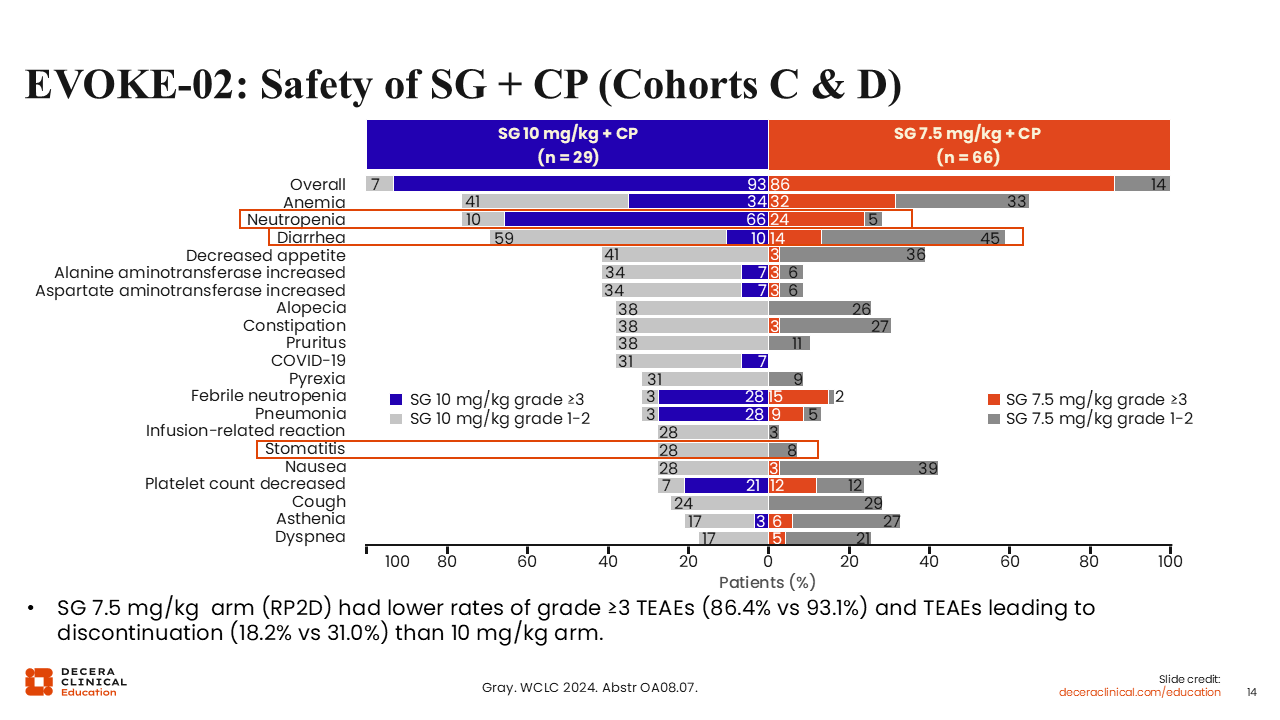
EVOKE-02: Safety of SG + CP (Cohorts C and D)
Benjamin Levy, MD, FASCO:
Rates of stomatitis were not as high with the SG combinations as with the Dato-DXd.6,9,10,12-14 Grade 1/2 stomatitis occurred in 28% of patients with a higher dose of SG and 8% with the lower dose.14 SG did have higher rates of neutropenia and diarrhea than Dato-DXd regimens. Although these drugs have similar functions, we see different toxicity profiles.
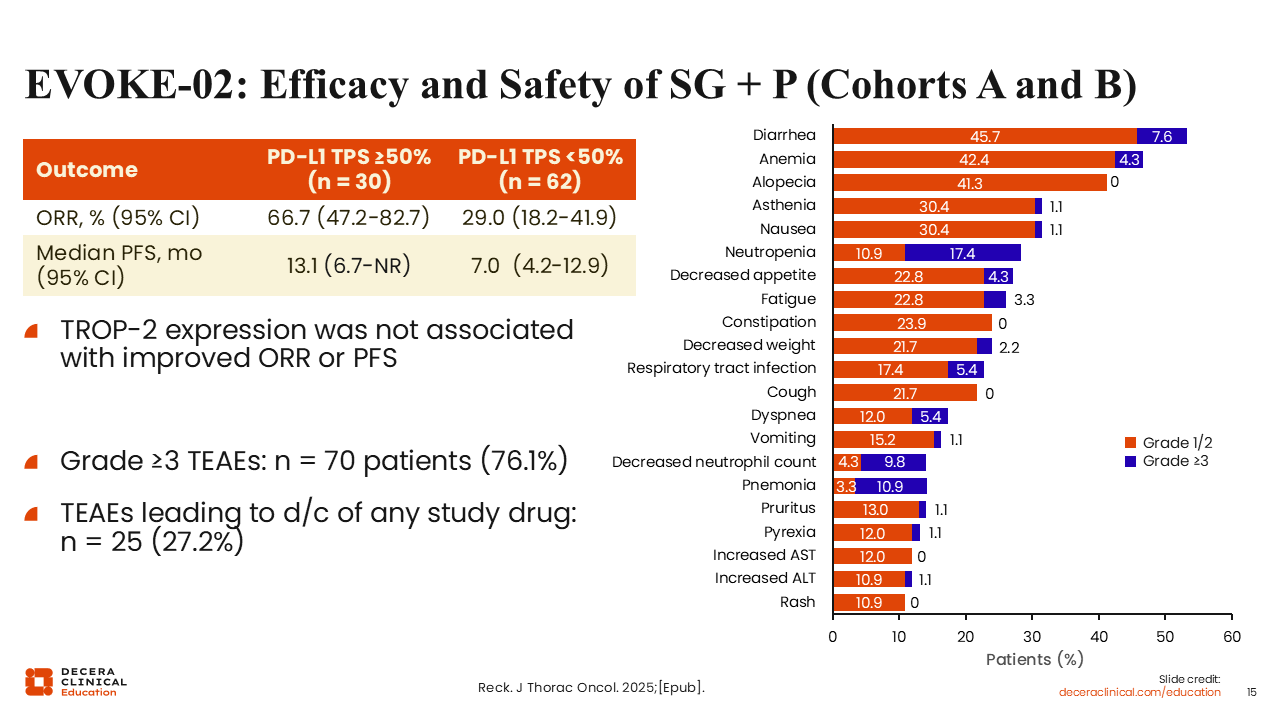
EVOKE-02: Efficacy and Safety of SG + P (Cohorts A and B)
Benjamin Levy, MD, FASCO:
In cohorts A and B that evaluated SG with pembrolizumab, ORR was more than double in patients with PD-L1–high (PD-L1 TPS ≥50%) disease vs those with PD-L1 TPS <50% (66.7% vs 29.0%).15 Median PFS was 13.1 vs 7.0 months. However, outcomes were not correlated with TROP-2 expression levels. Safety outcomes were similar to the cohorts C and D.
Sacituzumab Tirumotecan in EGFR-Mutated NSCLC
Benjamin Levy, MD, FASCO:
In December 2024, because of promising results of phase I/II and phase II single-arm trials, sac-TMT was granted breakthrough therapy designation by the FDA for the treatment of patients with advanced or metastatic nonsquamous NSCLC with EGFR exon 19 deletion or exon 21 L858R with progressive disease on or after TKI and platinum-based CT.16,17 Sac-TMT has now been further studied in 2 randomized trials in EGFR-mutated NSCLC.
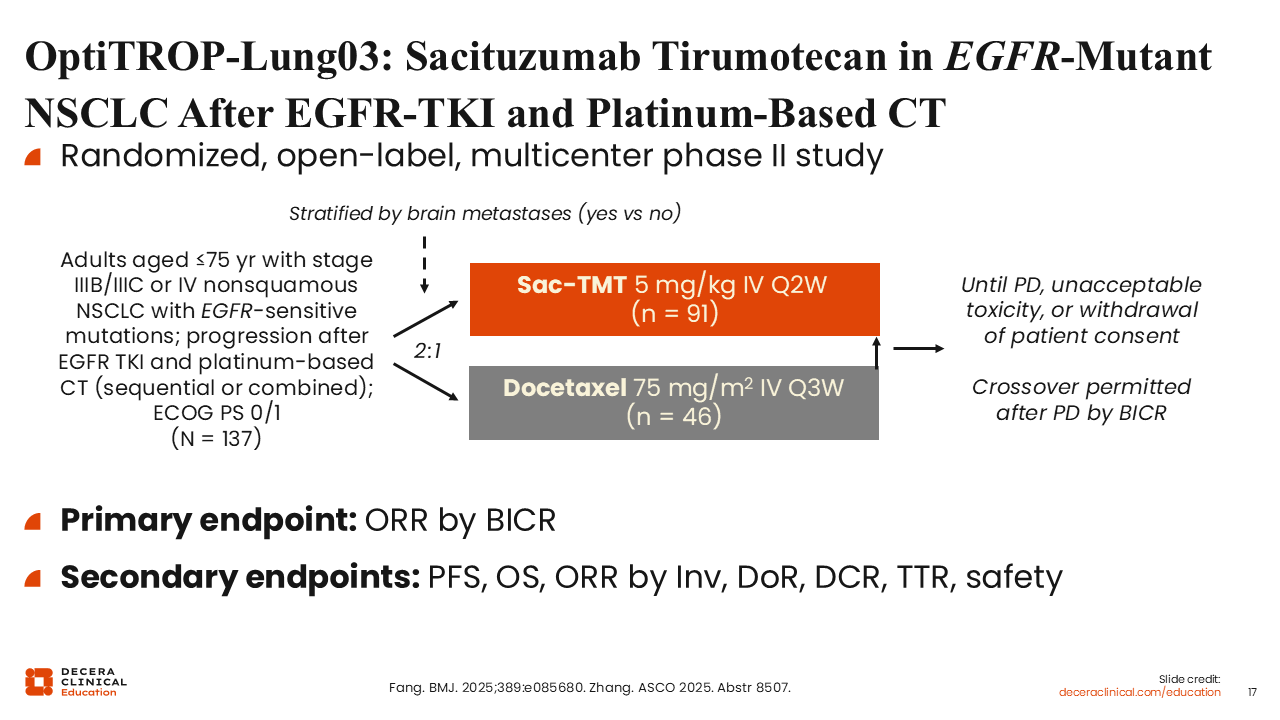
OptiTROP-Lung03: Sacituzumab Tirumotecan in EGFR-Mutant NSCLC After EGFR-TKI and Platinum-Based CT
Benjamin Levy, MD, FASCO:
OptiTROP-Lung03 was a randomized phase II study of sac-TMT vs docetaxel in patients with advanced nonsquamous NSCLC and EGFR-sensitive mutations.18,19 Patients were previously treated with both an EGFR TKI and platinum-based CT (sequentially or in combination). Crossover was permitted and primary endpoint was ORR by blinded independent central review (BICR). In total, 137 participants were enrolled and randomized at a 2:1 ratio (sac-TMT: n = 91; docetaxel: n = 46). Approximately one half (53%) of patients were treated on study in the third line, with 37% treated in later lines and just 10% in the second line.
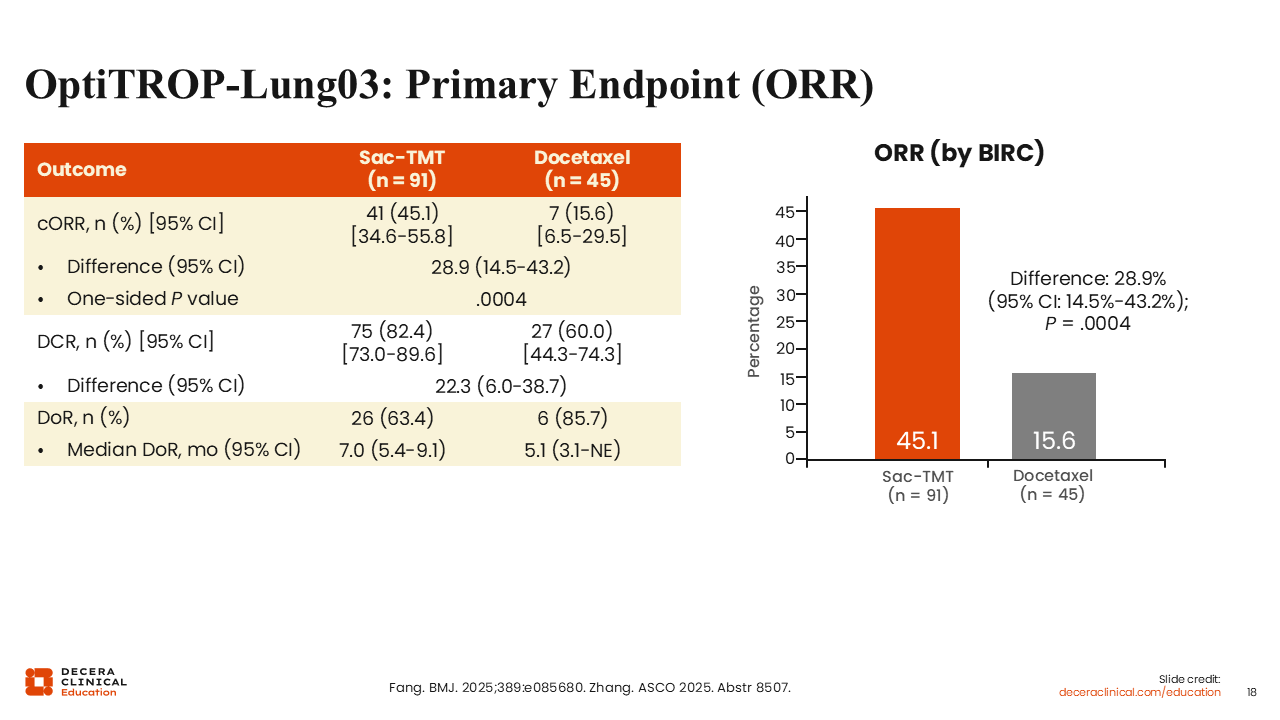
OptiTROP-Lung03: Primary Endpoint (ORR)
Benjamin Levy, MD, FASCO:
This study met its primary endpoint of a statistically superior ORR with sac-TMT vs docetaxel (45.1% vs 15.6%; P = .0004).18,19
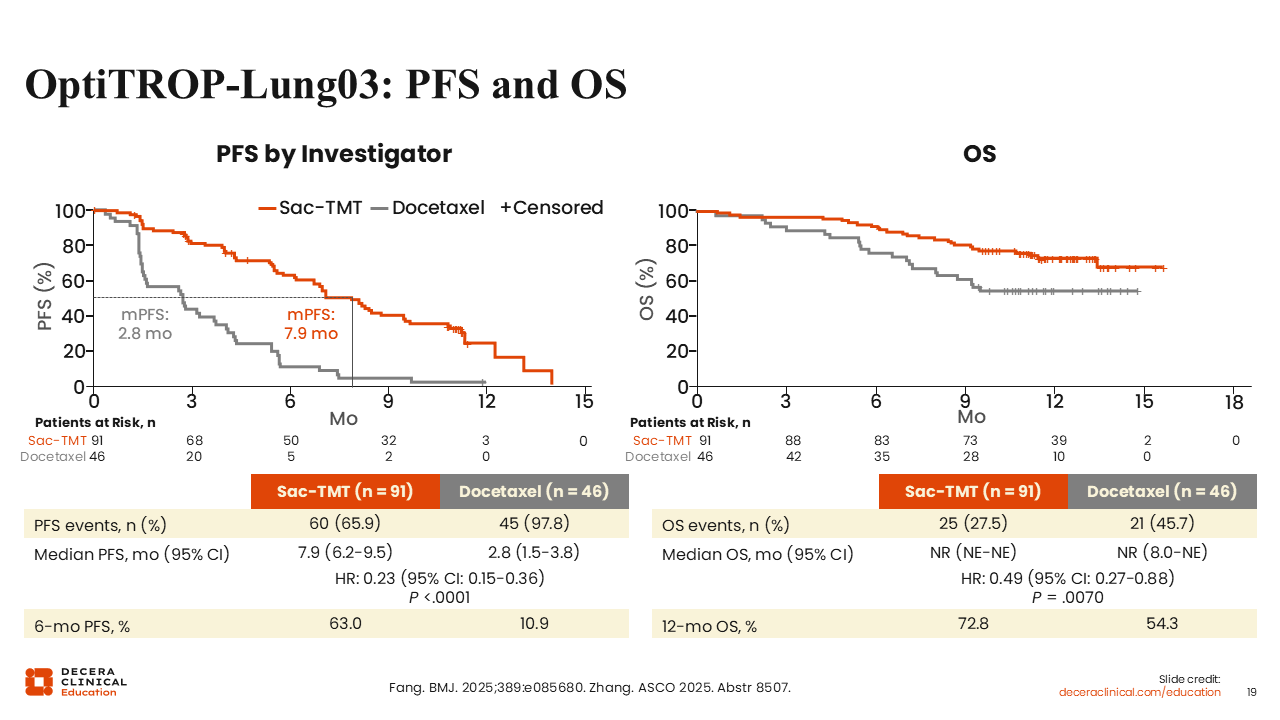
OptiTROP-Lung03: PFS and OS
Benjamin Levy, MD, FASCO:
Survival results were encouraging. Median PFS was 7.9 vs 2.8 months (HR: 0.23; P < .0001). 12-month OS rate was 72.8% vs 54.3% (HR: 0.49; P = .0070).18,19
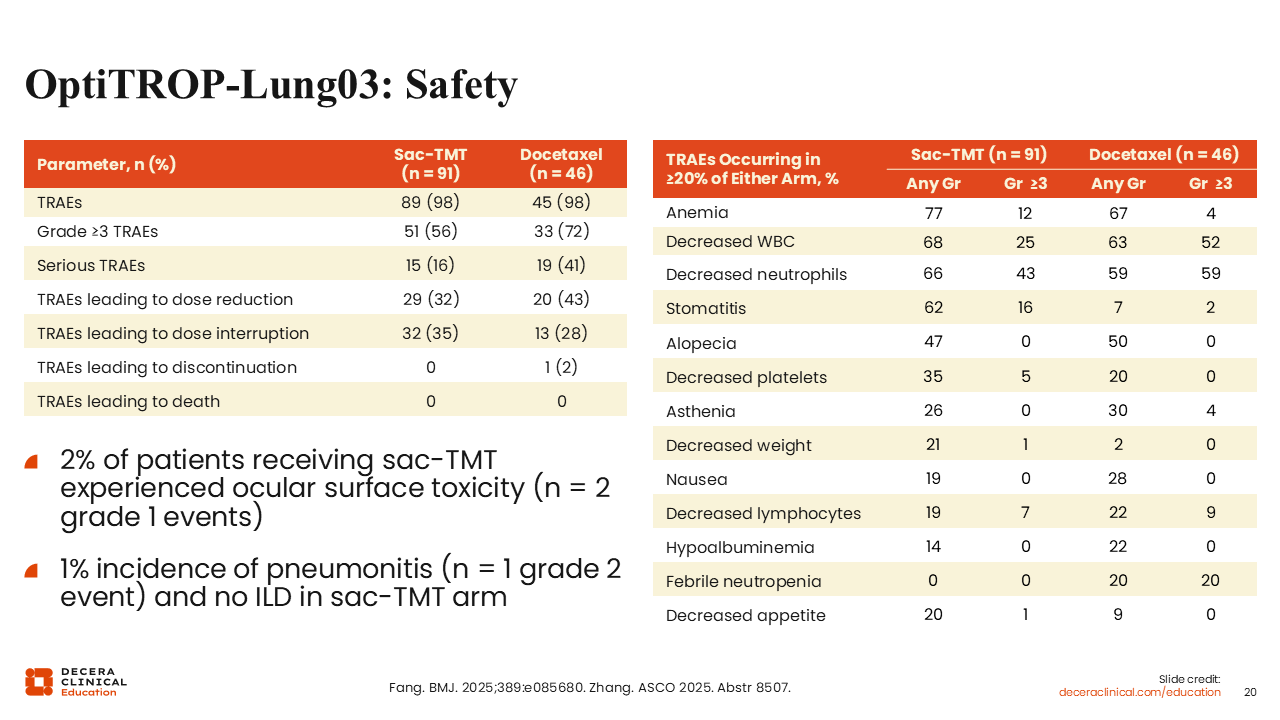
OptiTROP-Lung03: Safety
Benjamin Levy, MD, FASCO:
Like SG, the sac-TMT had high rates of decreased neutrophils (66%) and stomatitis (62%).18,19 Although decreased neutrophils occurred at a similar rate in the sac-TMT and the docetaxel arms (66% vs 59%; grade ≥3: 43% vs 59%). Ocular surface toxicity and pneumonitis were rare and low grade.
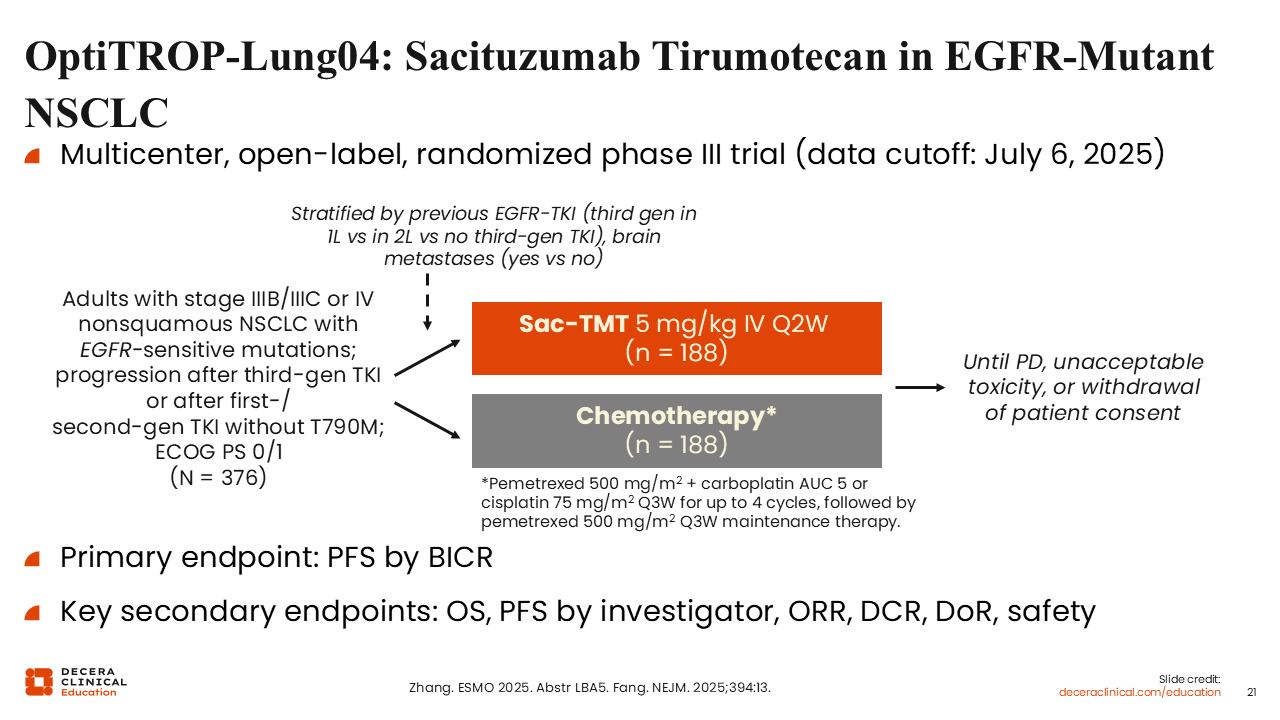
OptiTROP-Lung04: Sacituzumab Tirumotecan in EGFR-Mutant NSCLC
Benjamin Levy, MD, FASCO:
Similarly, the phase III trial OptiTROP-Lung04 randomized a larger patient population to sac-TMT and platinum-based CT with pemetrexed.20,21 Participants also had advanced nonsquamous NSCLC with EGFR-sensitive mutations and progression after a third-generation TKI (or second-generation TKI if they had T790M mutations). In total, 376 patients were randomized 1:1 to each arm (n = 188 each). The study’s primary endpoint was PFS by BICR.
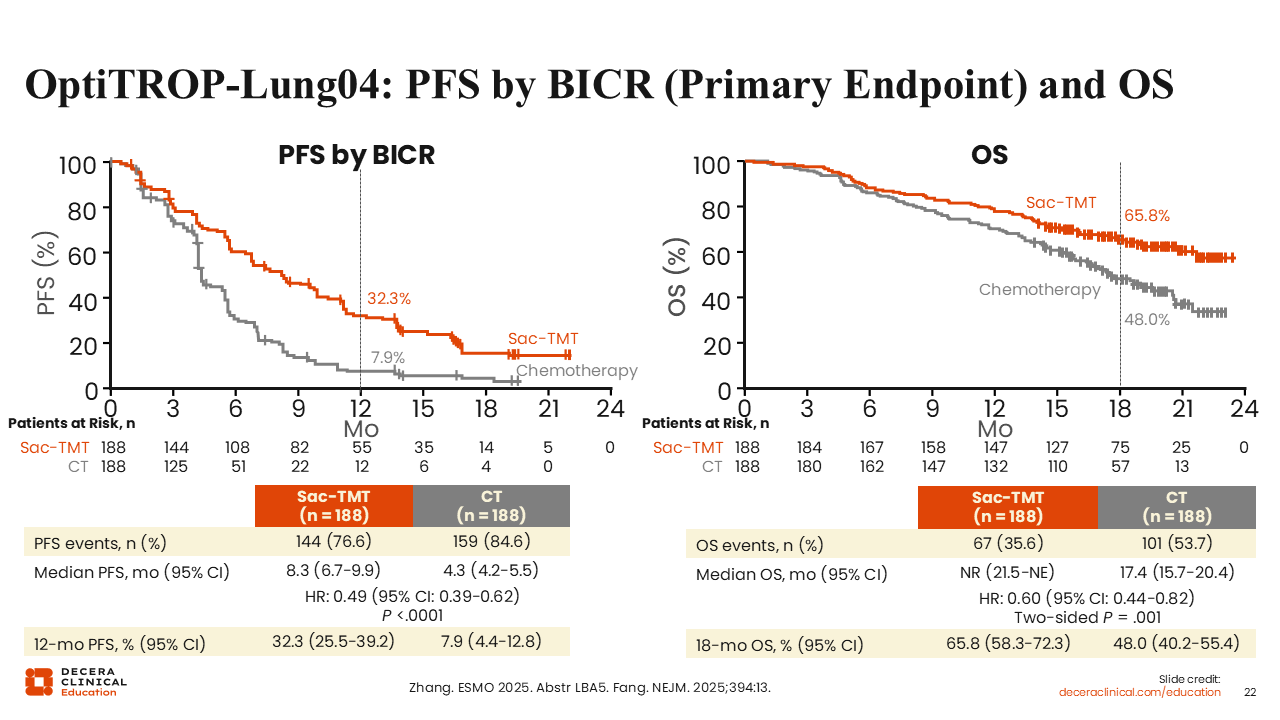
OptiTROP-Lung04: PFS by BICR (Primary Endpoint)
Benjamin Levy, MD, FASCO:
As reported at the 2025 European Society for Medical Oncology Congress (ESMO), sac-TMT showed statistically superior PFS vs CT with median PFS of 8.3 vs 4.3 months (HR: 0.49; P <.0001).20,21 OS data were immature but also showed improvement with sac-TMT (HR: 0.60; P = .001). OS rates at 18 months were 65.8% vs 48.0%.
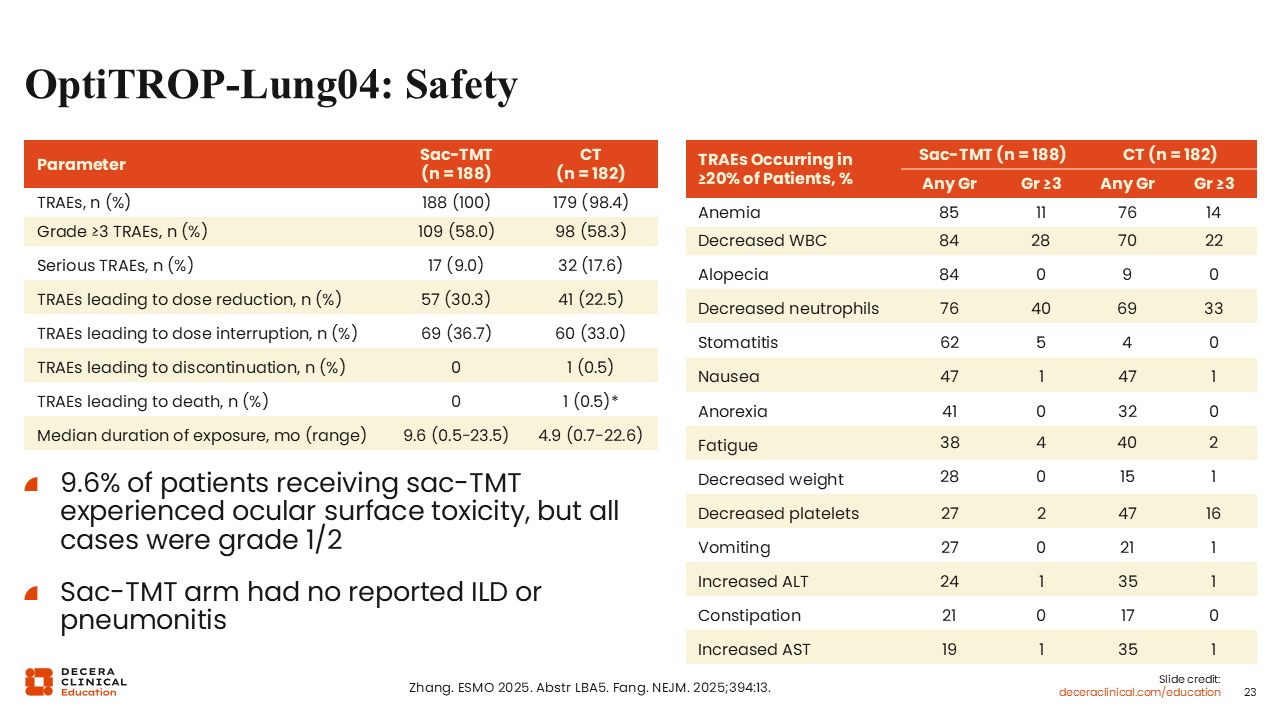
OptiTROP-Lung04: Safety
Benjamin Levy, MD, FASCO:
Safety results were similar to the OptiTROP-Lung03 data with a slightly higher rate of ocular surface toxicity of 9.6%.20,21
Select Phase III Trials of Anti–TROP-2 ADCs in NSCLC
Benjamin Levy, MD, FASCO:
Treatment of EGFR-positive NSCLC has recently become incredibly complex, but Dato-DXd has a clear role after the FLAURA2 regimen of osimertinib plus CT.22-24 This agent is a promising option for our patients, particularly if there are no available clinical trials.
Looking to the future, I am excited to see the results of the phase III TROPION-Lung08 study comparing pembrolizumab with or without Dato-DXd for PD-L1–high advanced, nonsquamous NSCLC (NCT05215340). We also need larger studies to confirm the promising results of SG and sac-TMT in NSCLC.