CME
Congress to Clinic: Expert Perspectives on Therapeutic Advances and Practice Changing Data Emerging From the 2025 EHA and ICML Congresses
Physicians: Maximum of 1.00 AMA PRA Category 1 Credit™
Released: September 25, 2025
Expiration: March 24, 2026
Activity
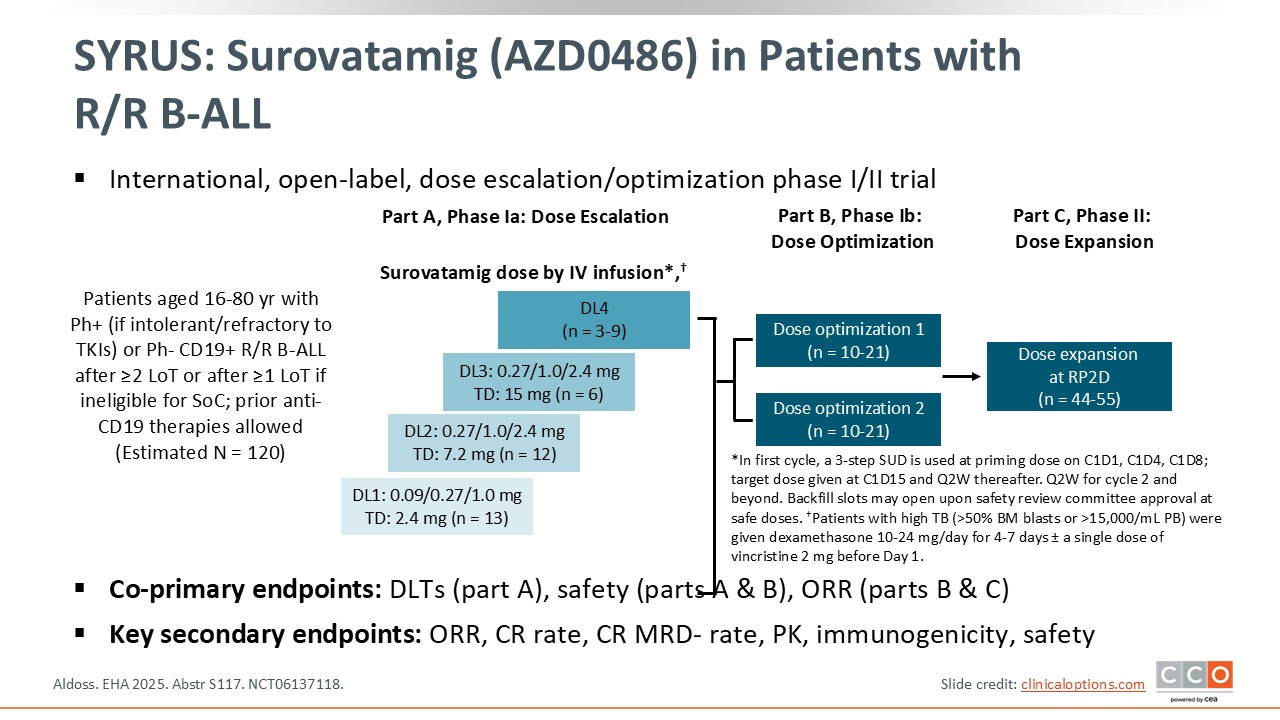
SYRUS: Surovatamig (AZD0486) in Patients With R/R B-ALL
Max S. Topp, MD:
Surovatamig, or AZD0486, is a CD19 x CD3 bispecific antibody that engages T‑cells by targeting CD3 and CD19 that is being investigated in patients with B-ALL in the phase I/II SYRUS trial. Enrolled patients had received ≥2 prior lines of therapy (≥1 if ineligible for standard of care), most of whom were exposed to blinatumomab, CAR T-cells, or both, and experienced relapse. Four different doses of surovatamig were administered intravenously. The coprimary endpoints were dose-limiting toxicities, safety, and ORR. Key secondary endpoints included the CR rate, pharmacokinetics, and safety. This trial consists of 3 parts, and EHA 2025 reported the dose‑finding results.1
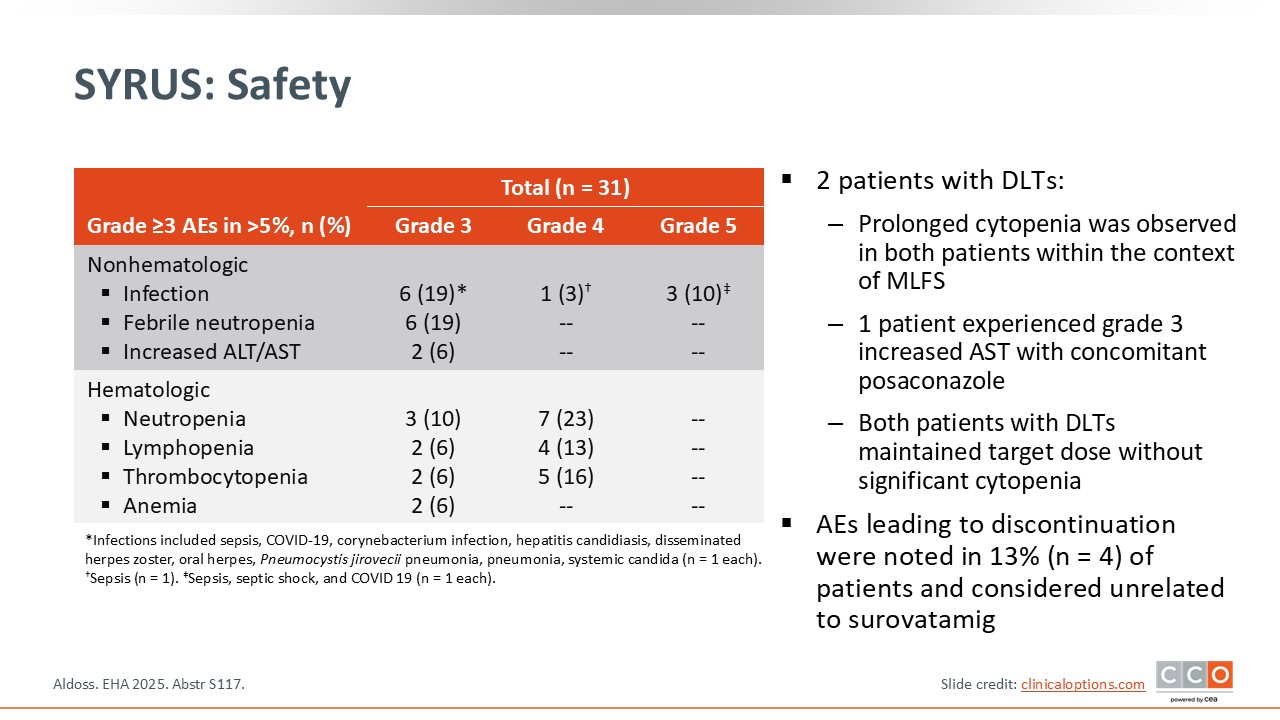
SYRUS: Safety
Max S. Topp, MD:
At EHA 2025, results from 31 patients were presented. Regarding adverse events (AEs), investigators noted grade 3 infections, as expected in this patient group: neutropenia, increased alanine aminotransferase/aspartate aminotransferase levels, and hematologic toxicities.1
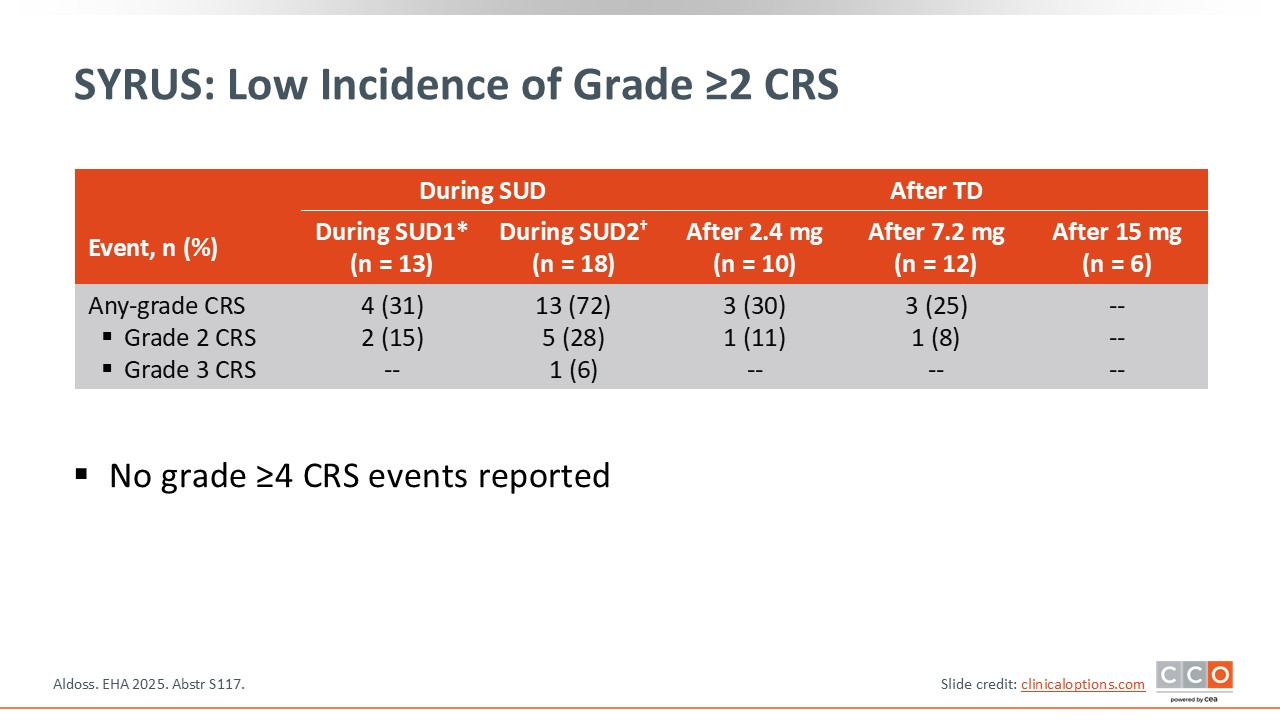
SYRUS: Low Incidence of Grade ≥2 CRS
Max S. Topp, MD:
Cytokine-release syndrome (CRS) occurred but was mitigated by the protocol use of step‑up dosing; the incidence of grade ≥2 CRS was low, and no grade ≥4 CRS events were reported.1
Of importance, immune effector cell–associated neurotoxicity syndrome (grade 3) was noted in only 1 of 31 patients.
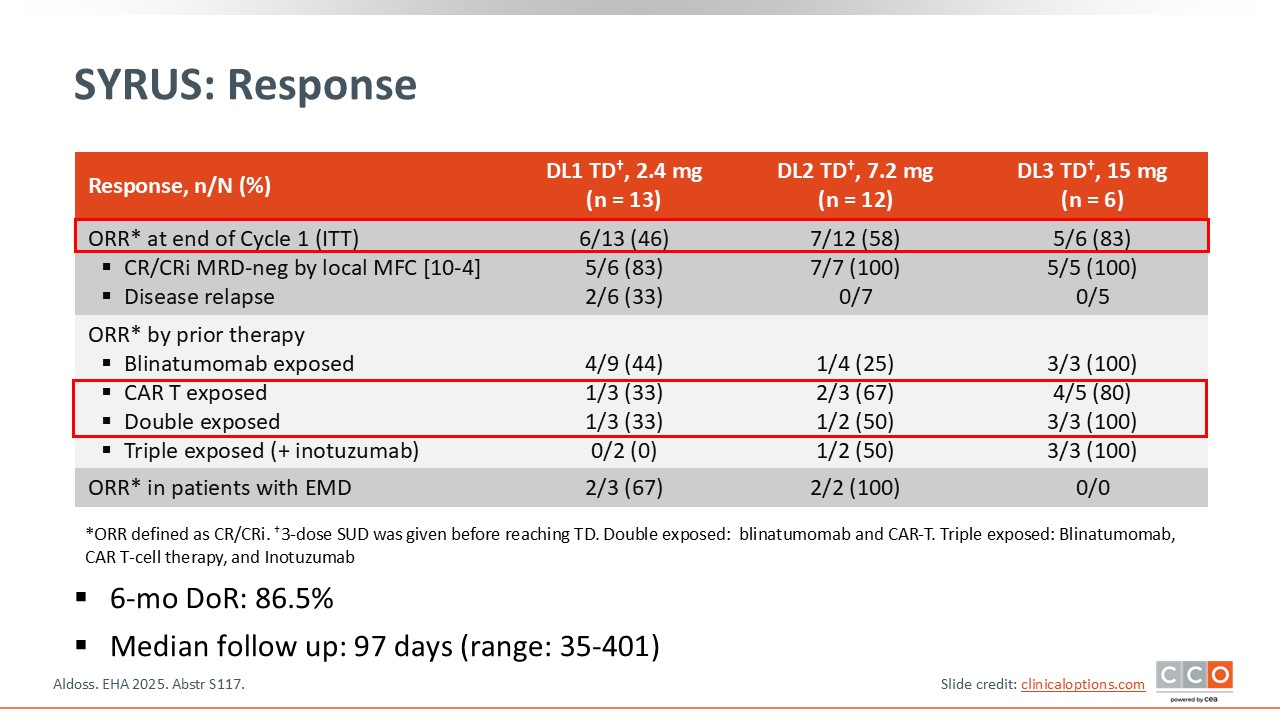
SYRUS: Response
Max S. Topp, MD:
In the DL3 cohort, 5 of 6 patients (83%) achieved CR/CRi, and all 5 assessed were MRD‑negative. Among 3 patient exposed to blinatumomab, all achieved CR, and among those exposed to CAR T-cell therapy, the CR rate was 80%. Among participants exposed to both blinatumomab and CAR T-cell therapy (double exposed), the CR rate was 100%, suggesting a highly active compound in this setting. The data also suggest that CD19 expression is maintained.1
SYRUS: Investigator's Conclusions
Max S. Topp, MD:
Overall, surovatamig showed a manageable safety profile and encouraging efficacy in heavily pretreated R/R B‑ALL. SYRUS is moving into phase II, which is very exciting.1
CaDAnCe-101: BGB-16673, a BTK degrader, in Patients with B-Cell Malignancies (CLL/SLL Cohort)
Pier Luigi Zinzani, MD, PhD:
CaDAnCe-101 is a phase I/II trial of BGB-16673, a BTK degrader, in patients with B-cell malignancies including chronic lymphocytic leukemia (CLL)/small lymphocytic lymphoma (SLL). At EHA 2025, preliminary data from part 1A (dose escalation) and part 1B (safety expansion) in the CLL/SLL cohort of patients were presented. The patients in this cohort had received ≥2 prior lines of therapy, including a covalent BTK inhibitor (BTKi), if approved for disease control. The coprimary endpoints for phase I were safety, tolerability, maximum tolerated dose (MTD), and recommended dose for expansion.2
CaDAnCe-101: Baseline Characteristics (CLL/SLL Cohort)
Pier Luigi Zinzani, MD, PhD:
A total of 66 patients with CLL/SLL and a median age of 70 years, were enrolled and treated. Approximately 38% had a BTK mutation, nearly 16% had a PLCG2 mutation, and approximately 8% presented with both. More than 65% of patients had del(17p) and/or TP53 mutation, and 50% had a complex karyotype. The median number of prior lines of therapy was 4, so patients were heavily pretreated, and more than 88% of patients had discontinued a prior BTKi because of disease progression.2
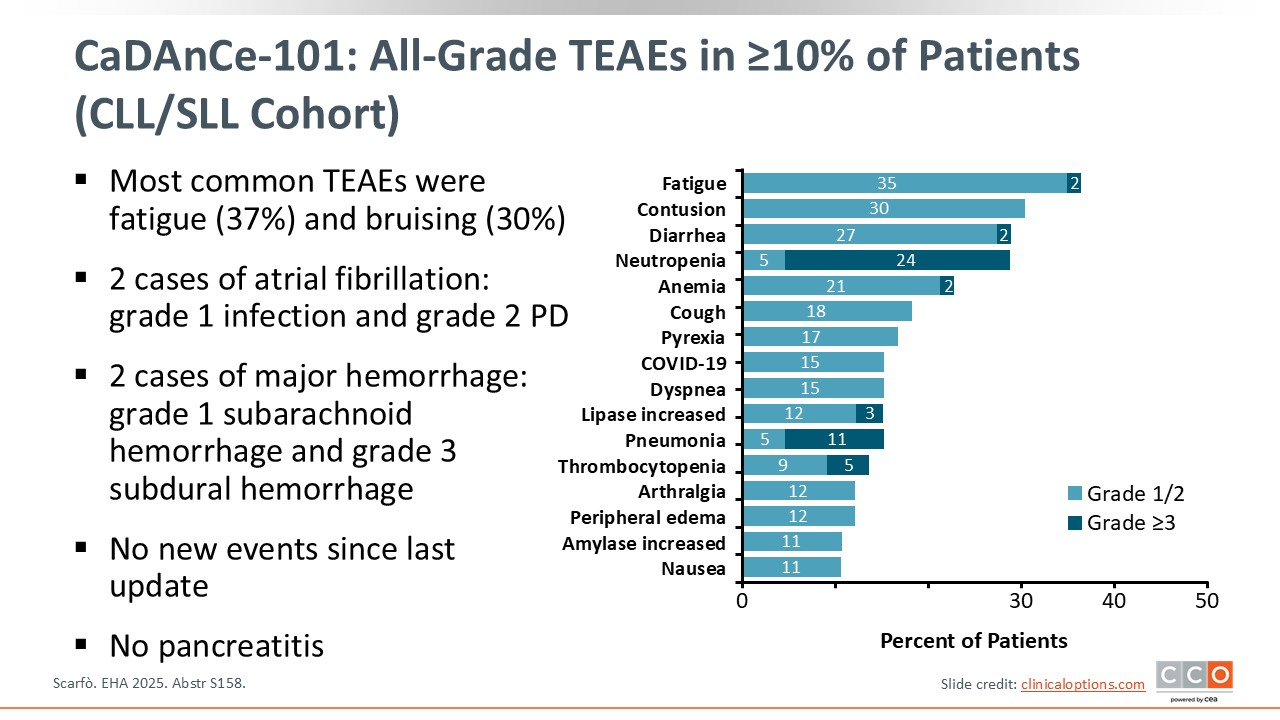
CaDAnCe-101: All-Grade TEAEs in ≥10% of Patients (CLL/SLL Cohort)
Pier Luigi Zinzani, MD, PhD:
The most common AEs were fatigue (37%) and contusion or bruising (30%). Atrial fibrillation was observed in only 2 patients, and pancreatitis was not observed. Major hemorrhage was also observed in only 2 patients. No new AEs occurred since the last update, and pancreatitis was not observed.2
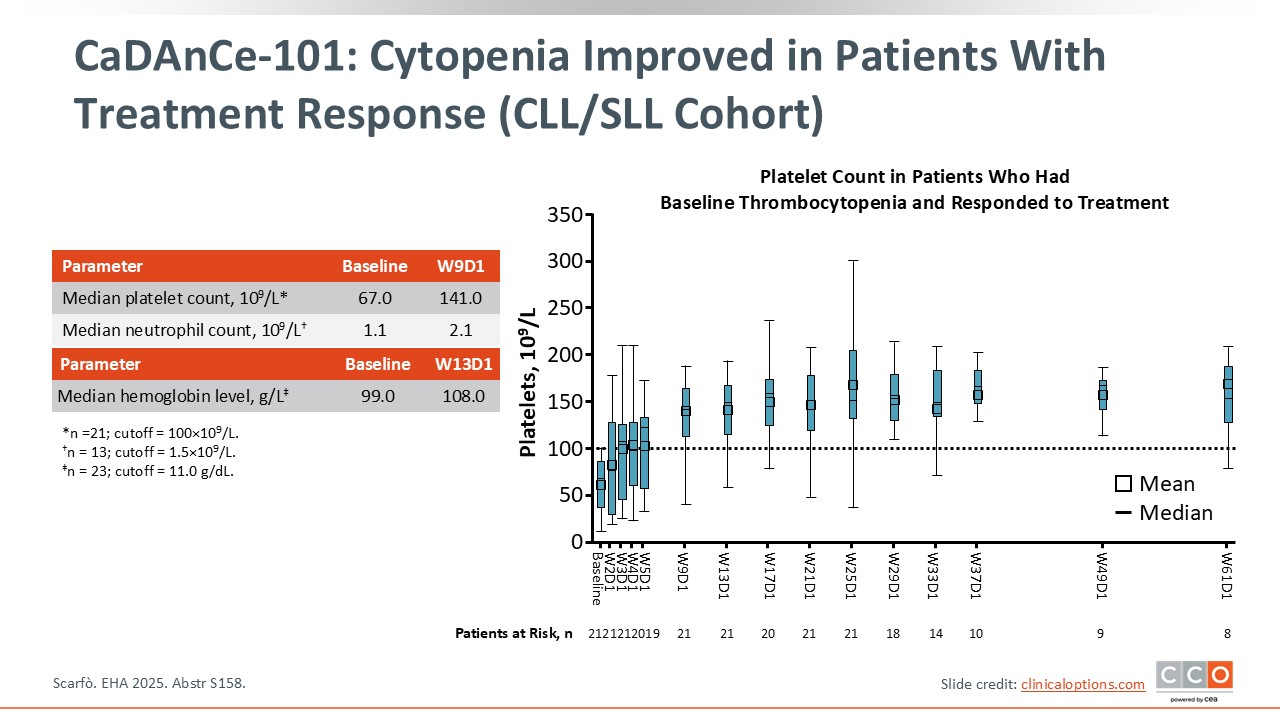
CaDAnCe-101: Cytopenia Improved in Patients With Treatment Response (CLL/SLL Cohort)
Pier Luigi Zinzani, MD, PhD:
Another important point in favor of BGB-16673 is the rapid and significant improvement in cytopenia that was observed. As shown here, there was a significant improvement in platelet counts as well as neutrophil counts and hemoglobin levels during the first weeks of treatment.2
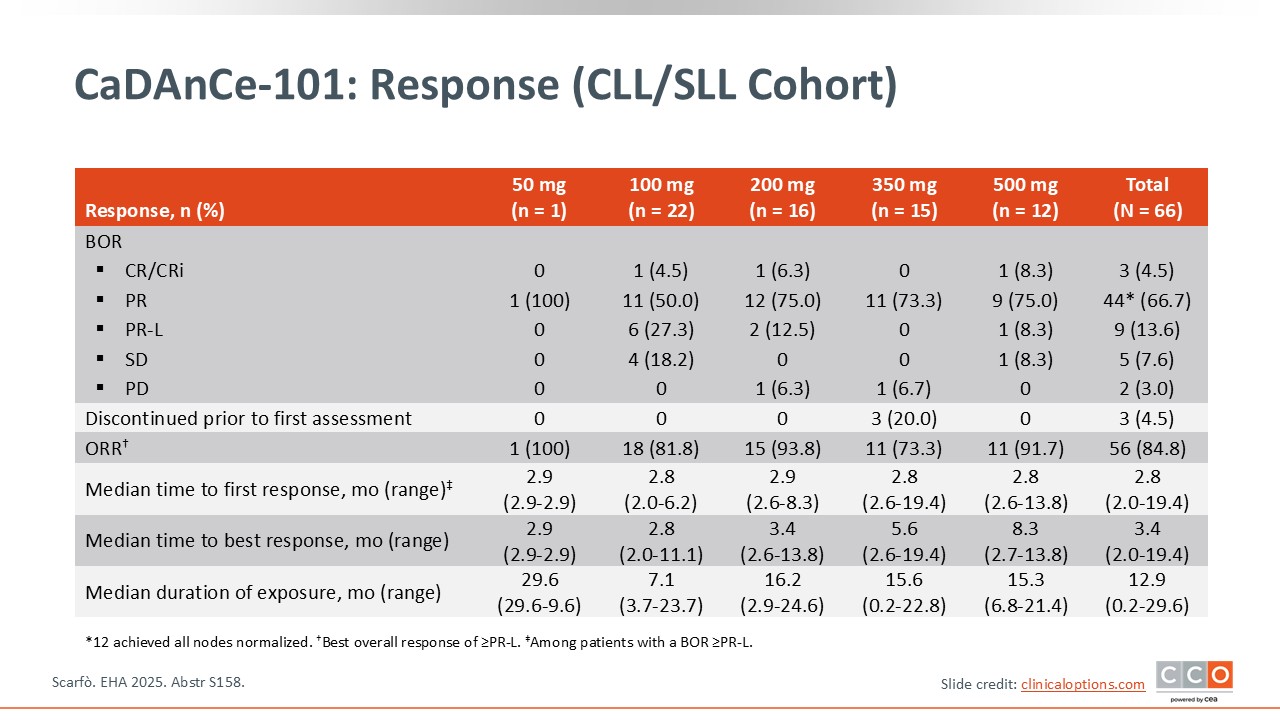
CaDAnCe-101: Response (CLL/SLL Cohort)
Pier Luigi Zinzani, MD, PhD:
According to the preliminary results from phase I, the ORR in the total population of patients with CLL/SLL was approximately 85%, with a CR rate of 4.5%. Among the 5 different doses, the best result in terms of a significant response was observed at the 200-mg dose level.2
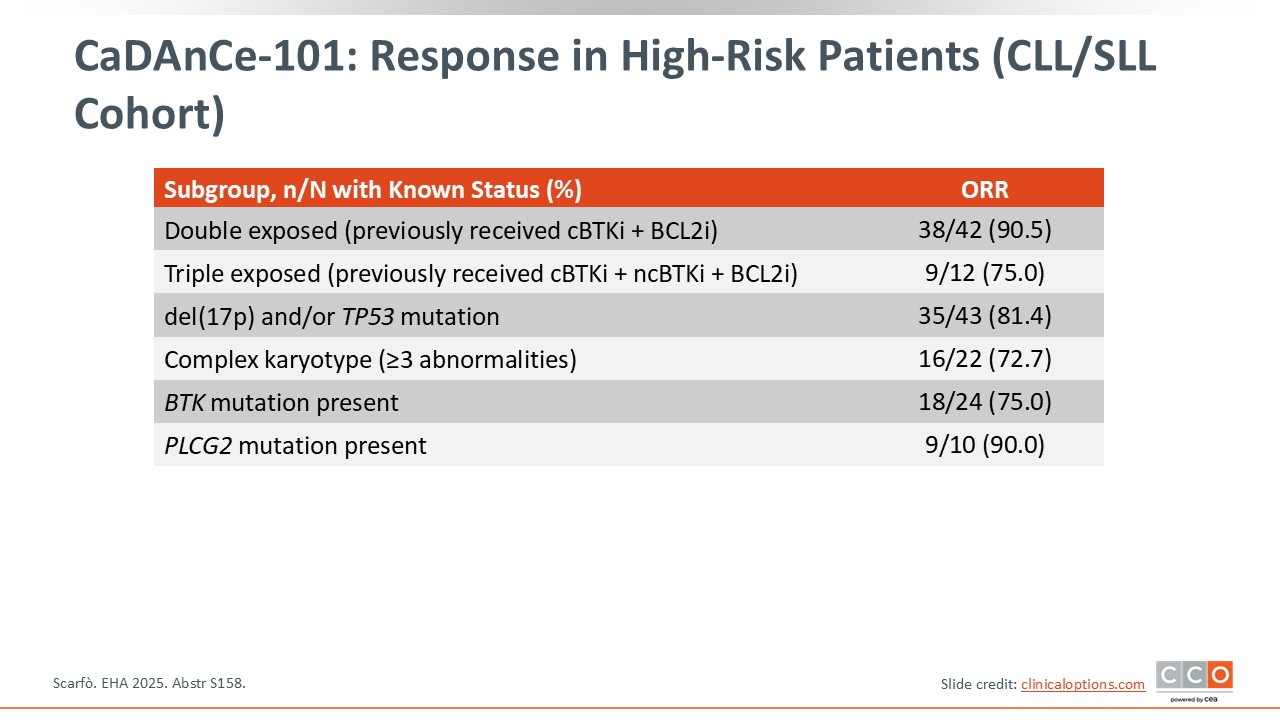
CaDAnCe-101: Response in High-Risk Patients (CLL/SLL Cohort)
Pier Luigi Zinzani, MD, PhD:
It is important to note that the ORRs were also high in high-risk patient subsets. The ORR for patients who were previously treated with both a covalent BTKi and BCL2 inhibitor was 90.5%. For patients who had received a prior covalent BTKi, noncovalent BTKi, and BCL2 inhibitor, the ORR was 75%. For patients with del(17p) and/or TP53 mutation, the ORR was >80%, and for patients with a complex karyotype, the ORR was >72%. Among patients with a BTK mutation and PLCG2 mutation, the ORRs were 75% and 90%, respectively.2
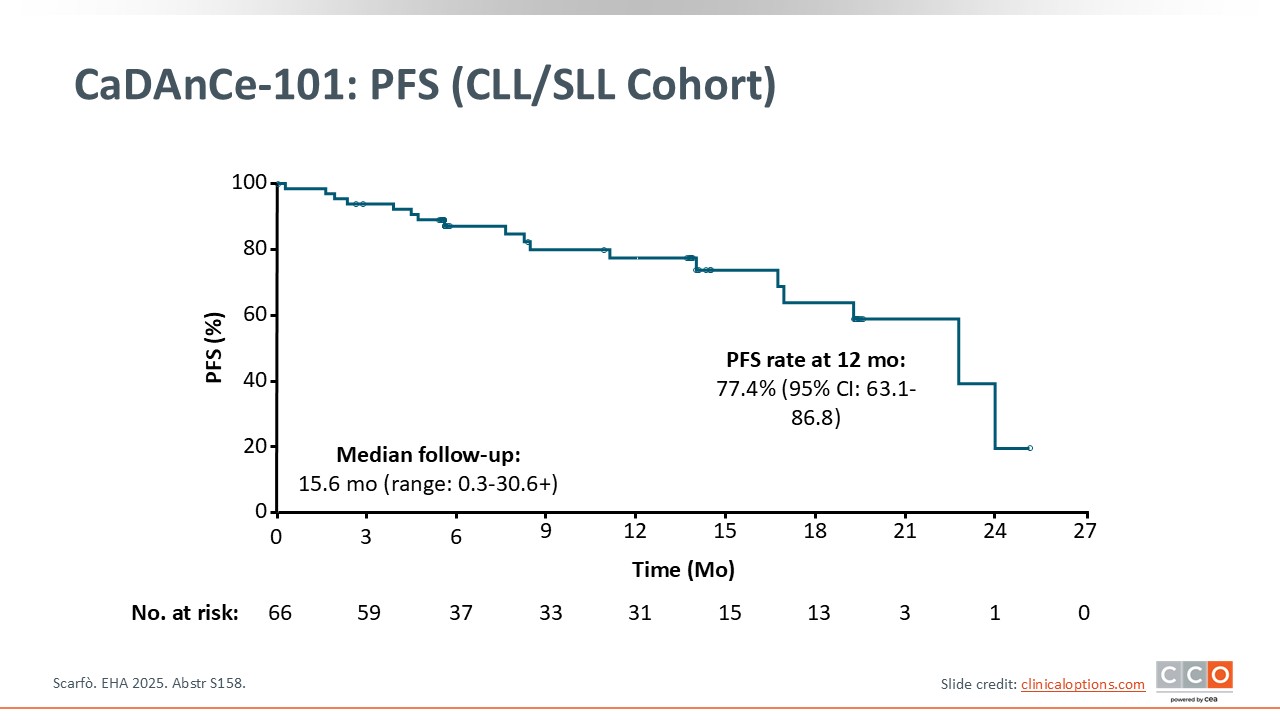
CaDAnCe-101: PFS (CLL/SLL Cohort)
Pier Luigi Zinzani, MD, PhD:
At a median follow‑up 15.6 months, the 12‑month PFS rate was 77.4% (95% CI: 63.1-86.8); longer follow‑up needed.2
CaDAnCe-101: Investigator's Conclusions (CLL/SLL Cohort)
Pier Luigi Zinzani, MD, PhD:
Preliminary phase I data from the cohort of patients with CLL/SLL enrolled in CaDAnCe-101 established an MTD of 200 mg. The safety profile was clinically manageable, and the ORRs were encouraging, especially in subsets with high-risk disease. Moreover, the median time to first response was <3 months.2 Overall, these early data suggest promising antitumor activity for this agent.
POLARGO: Polatuzumab Vedotin + R-GemOx in Patients With Transplant-Ineligible R/R DLBCL
Pier Luigi Zinzani, MD, PhD:
The phase III POLARGO study is investigating Pola-R-GemOx in patients with DLBCL, including DLBCL NOS or those with a history of transformation of indolent disease to DLBCL. All patients experienced relapse or were refractory after at least 1 prior line of treatment and were considered ineligible for transplant. In total, 255 patients were randomized to either the Pola-R-GemOx arm or the control R-GemOx arm. The primary endpoint was OS.3
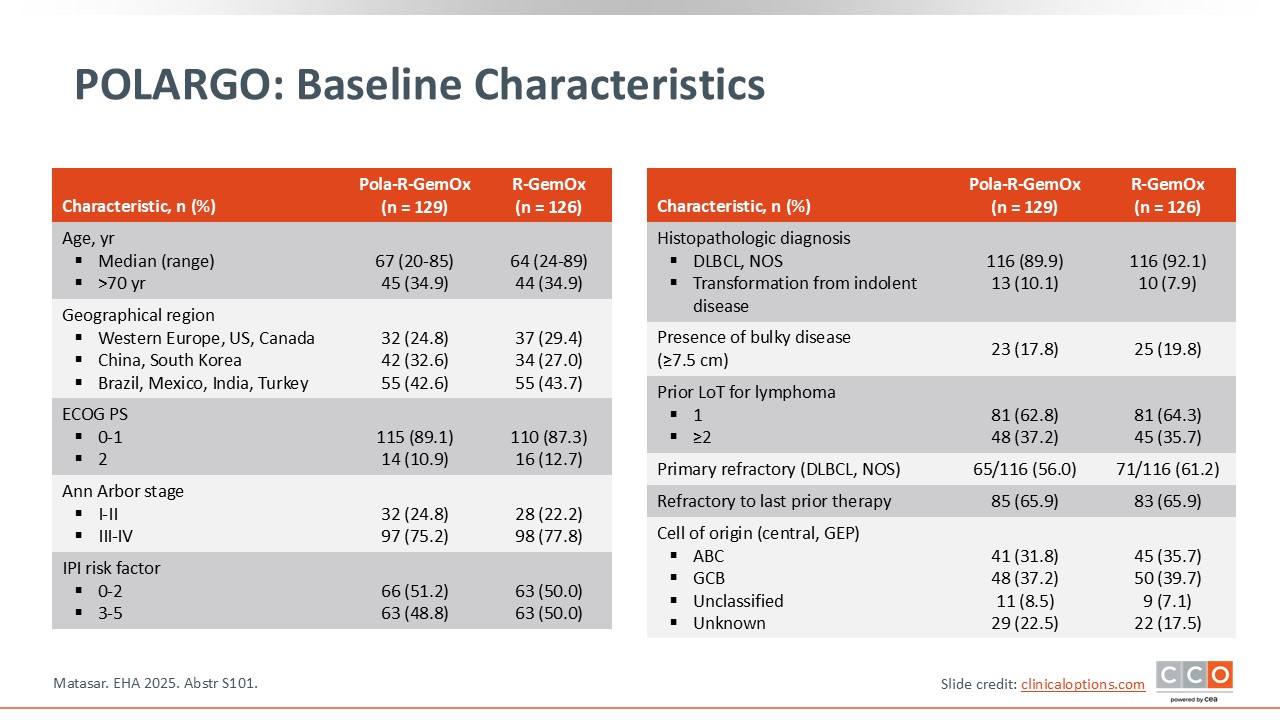
POLARGO: Baseline Characteristics
Pier Luigi Zinzani, MD, PhD:
There were no statistically significant differences in baseline characteristics between the 2 arms. Approximately 35% of patients were at least 70 years of age, with a median age of 67 and 64 years in the Pola-R-GemOx and R-GemOx arms, respectively. Bulky disease was noted in approximately 18% of patients, and more than 35% had received ≥2 prior lines of treatment. Two thirds of the patients were refractory to their last previous therapy.3
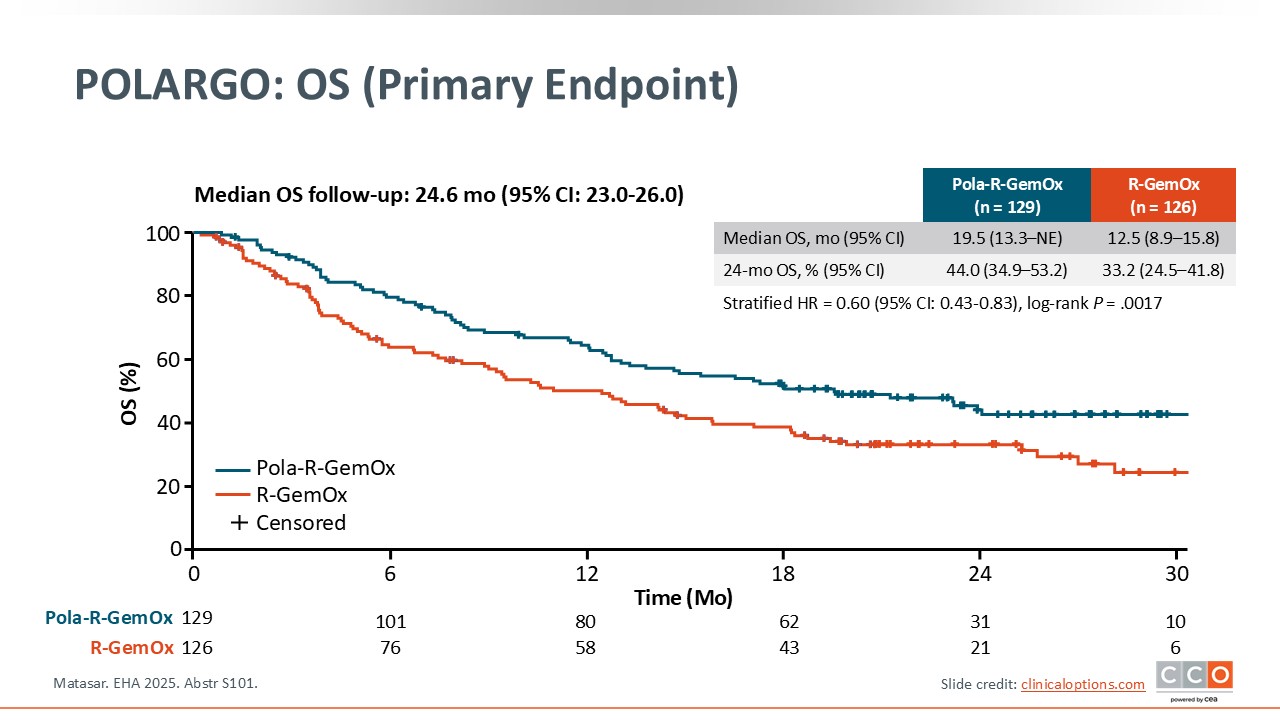
POLARGO: OS (Primary Endpoint)
Pier Luigi Zinzani, MD, PhD:
The primary endpoint of improved OS was met. The median OS was 19.5 months for Pola-R-GemOx vs 12.5 months for R-GemOx. The difference was statistically significant (HR: 0.60 (95% CI: 0.43-0.83, P = .0017). The OS rate at 2 years was 44% for the Pola-R-GemOx arm vs approximately 33% for the R-GemOx arm.3
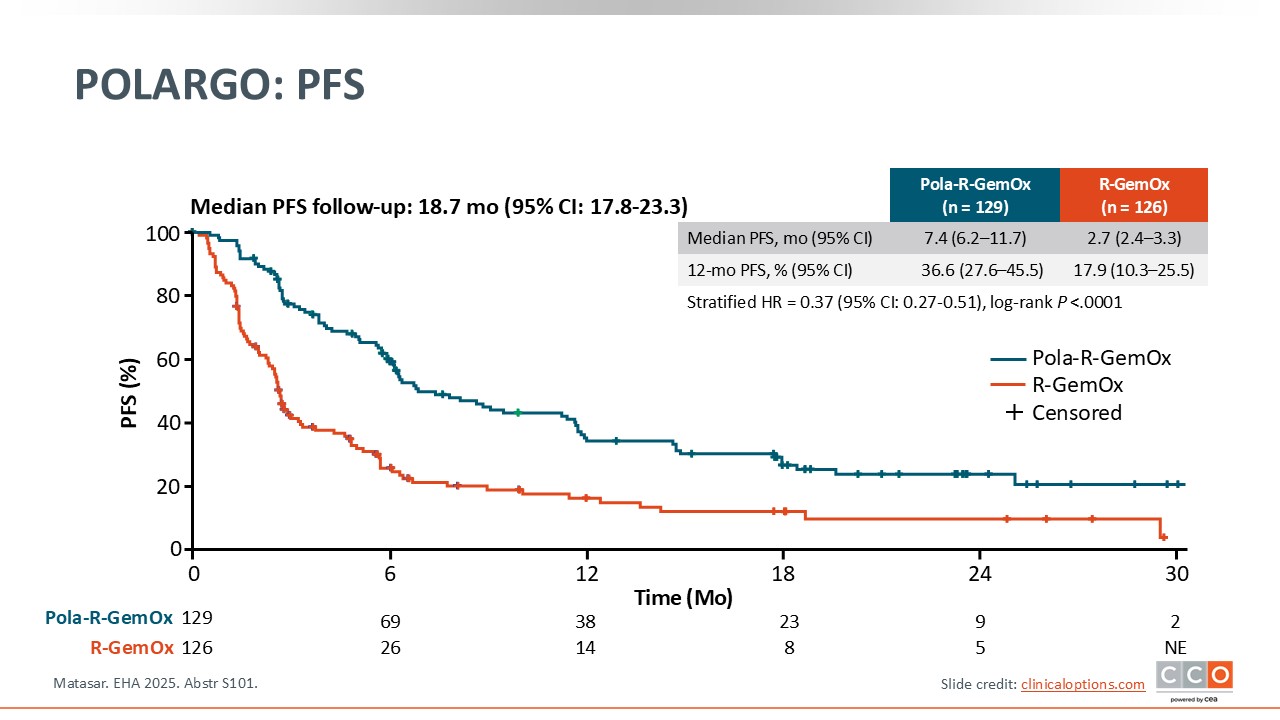
POLARGO: PFS
Pier Luigi Zinzani, MD, PhD:
PFS was a secondary endpoint in POLARGO. There was a statistically significant difference in favor of Pola-R-GemOx vs R-GemOx with a 63% risk reduction for disease progression or death (P <.0001). The median PFS was 7.4 months for Pola-R-GemOx vs 2.7 months for R-GemOx, and the 12-month PFS rates were 36.6% and 17.9%, respectively.3
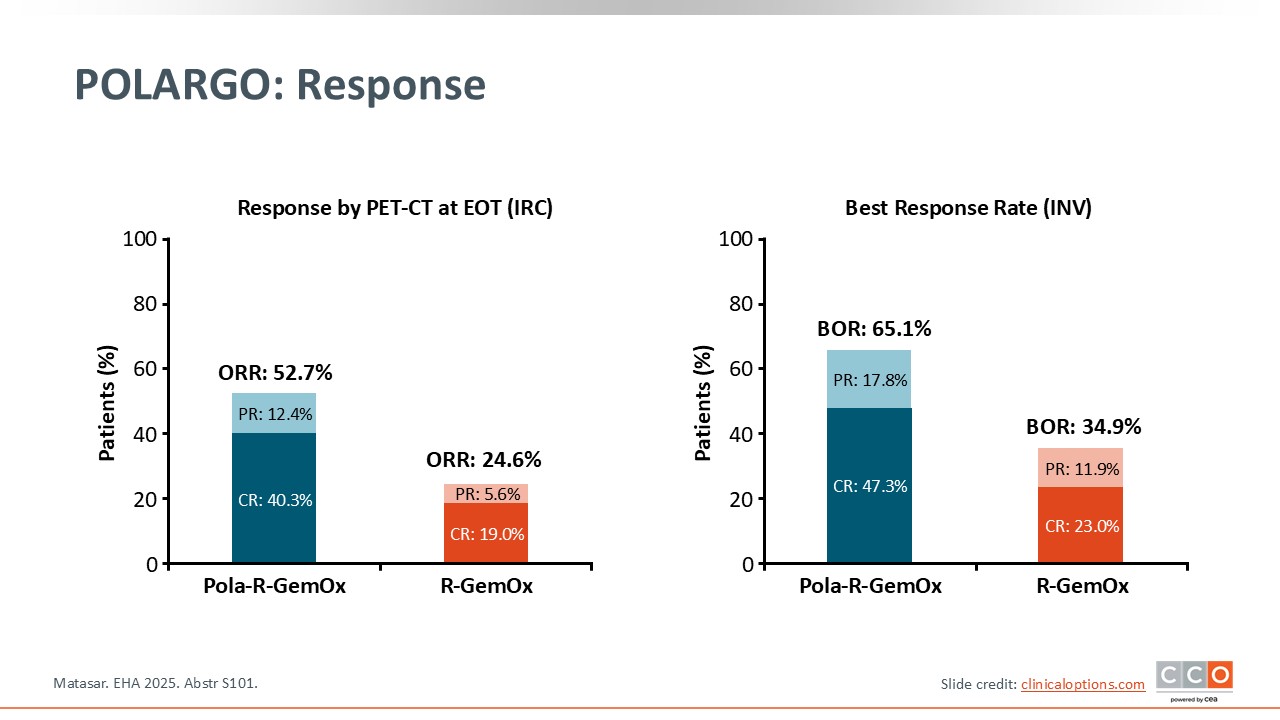
POLARGO: Response
Pier Luigi Zinzani, MD, PhD:
Similar results were observed in terms of disease response. The bar graph on the left represents the response rate according to the PET/CT evaluation at the end of treatment per independent central review. The ORR was approximately 53.0% for Pola-R-GemOx vs approximately 25.0% for R-GemOx, and the CR rate was 40.3% for Pola-R-GemOx vs 19.0% for R-GemOx. The bar graph on the right represents the best response rate per investigator for both arms, with Pola-R-GemOx also showing a similar benefit over R-GemOx alone.3
POLARGO: Subsequent Therapies
Pier Luigi Zinzani, MD, PhD:
More patients treated with R-GemOx required more subsequent treatments than those treated with Pola-R-GemOx. The percentage of patients who received at least 1 subsequent treatment was 45.7% for Pola-R-GemOx vs 65.1% for R-GemOx. The types of subsequent treatments after failure also differed between the 2 treatment arms.3
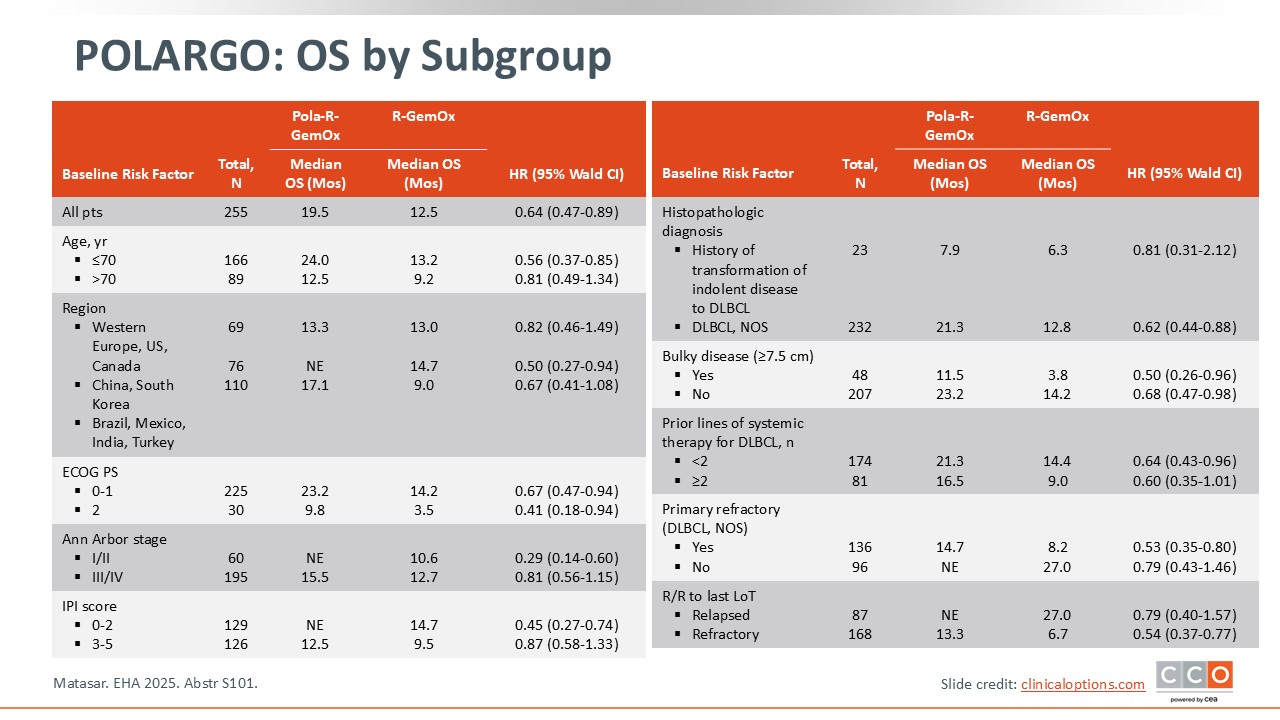
POLARGO: OS by Subgroup
Pier Luigi Zinzani, MD, PhD:
Regarding OS, the primary endpoint, the best results were obtained with Pola-R-GemOx regardless of baseline risk factors, including Ann Arbor stage, International Prognostic Index score, presence of bulky disease, and number of prior lines of treatment. These results were also interesting in patients with primary refractory disease.3
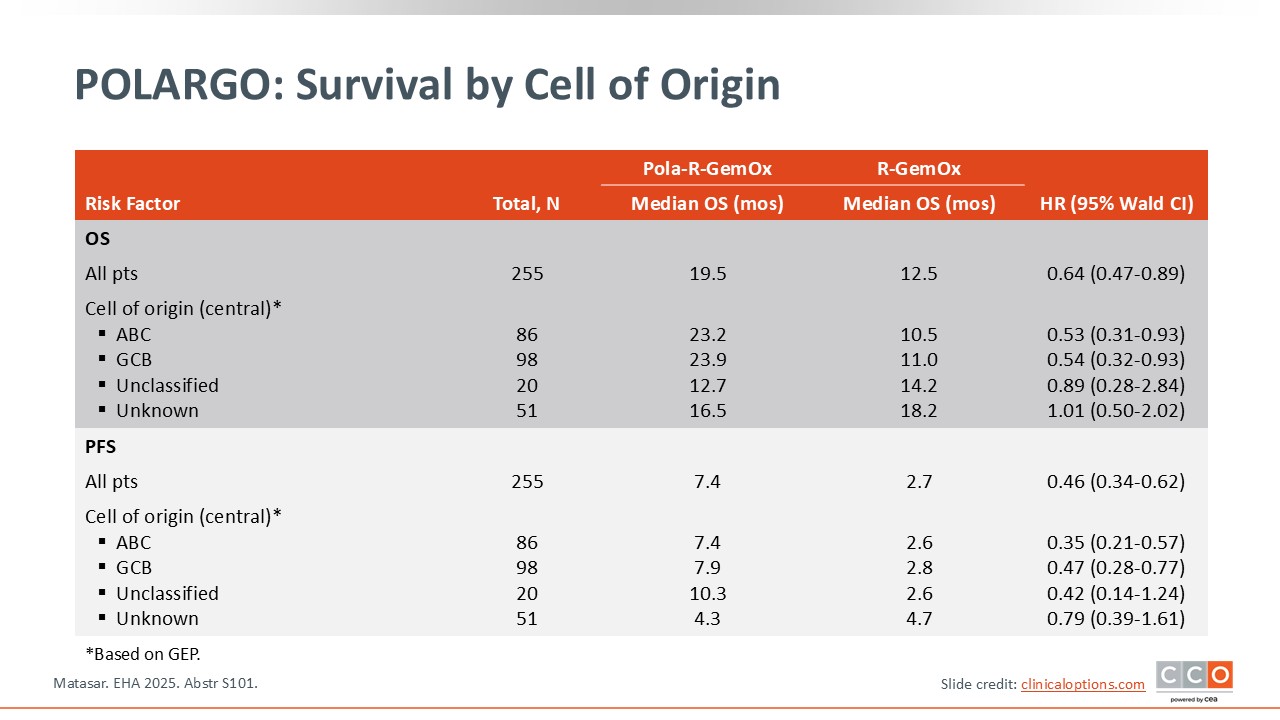
POLARGO: Survival by Cell of Origin
Pier Luigi Zinzani, MD, PhD:
Of importance, survival benefits were also seen regardless of the cell of origin. Regardless of whether the disease originated from activated B-cells or germinal center B-cell subtypes, the advantage with Pola-R-GemOx was superior to R-GemOx in terms of not only OS, but also PFS.3
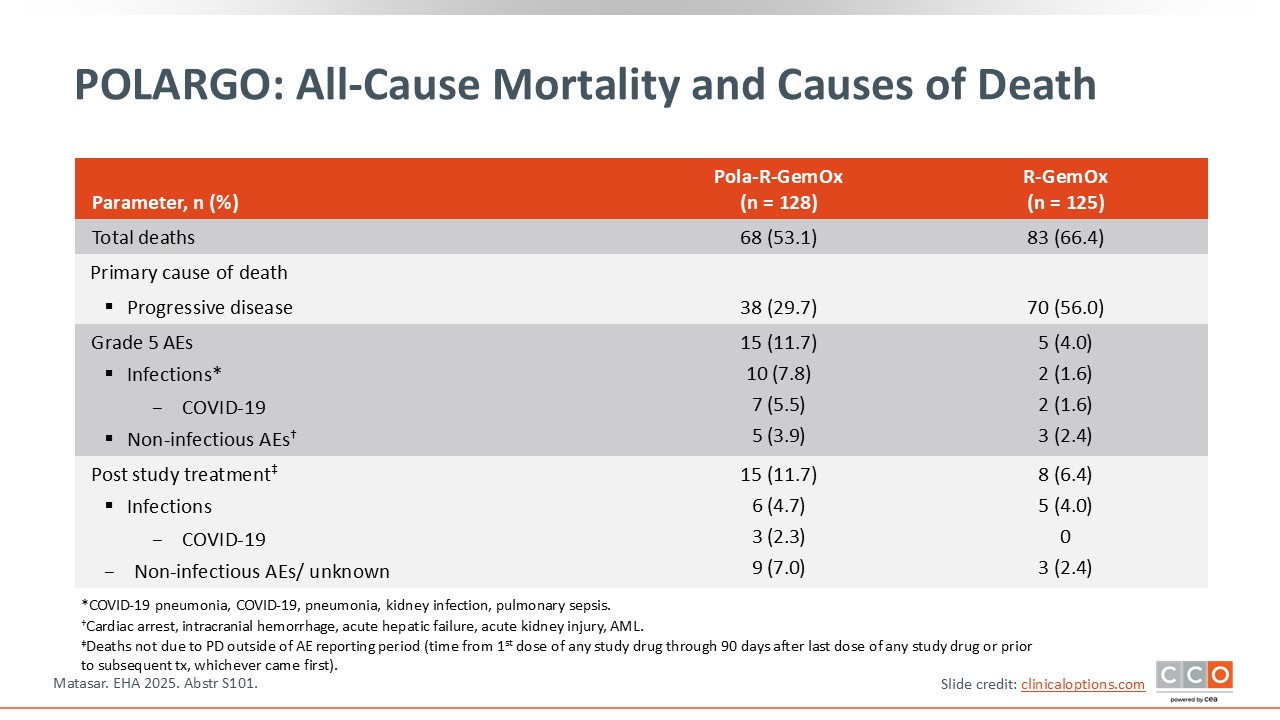
POLARGO: All-Cause Mortality and Causes of Death
Pier Luigi Zinzani, MD, PhD:
Overall mortality was because of disease progression and higher with R-GemOx (66.4%) than with Pola-R-GemOx (53.1%). In particular, the primary cause of death was progressive disease in nearly 30% of patients treated with Pola-R-GemOx and 56% in patients treated with R-GemOx.3
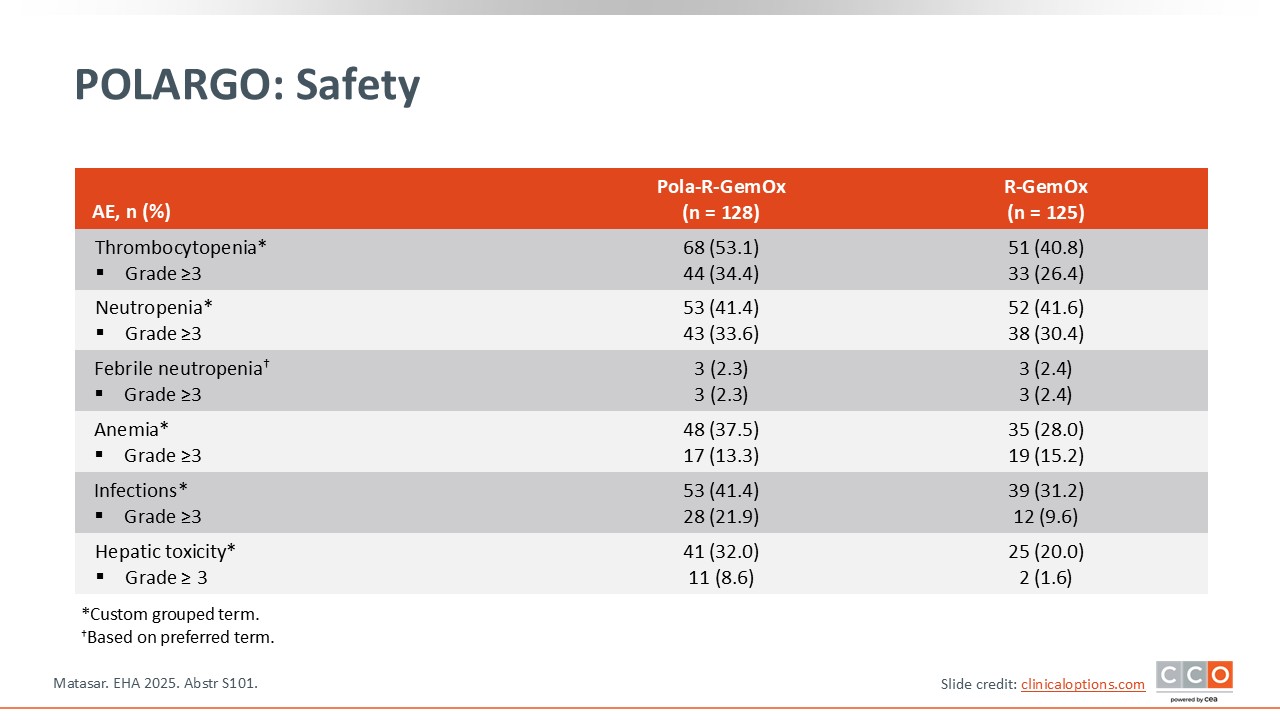
POLARGO: Safety
Pier Luigi Zinzani, MD, PhD:
In terms of safety, the most important AEs were hematologic and included grade ≥3 thrombocytopenia (34.4% vs 26.4%), neutropenia (33.6% vs 30.4%), febrile neutropenia (2.3% vs 2.4%), and anemia (13.3% vs 15.2%). In terms of hepatic toxicity, the percentages of patients experiencing any-grade hepatic toxicity were 32% in the Pola-R-GemOx arm and 20% in the R-GemOx arm.3
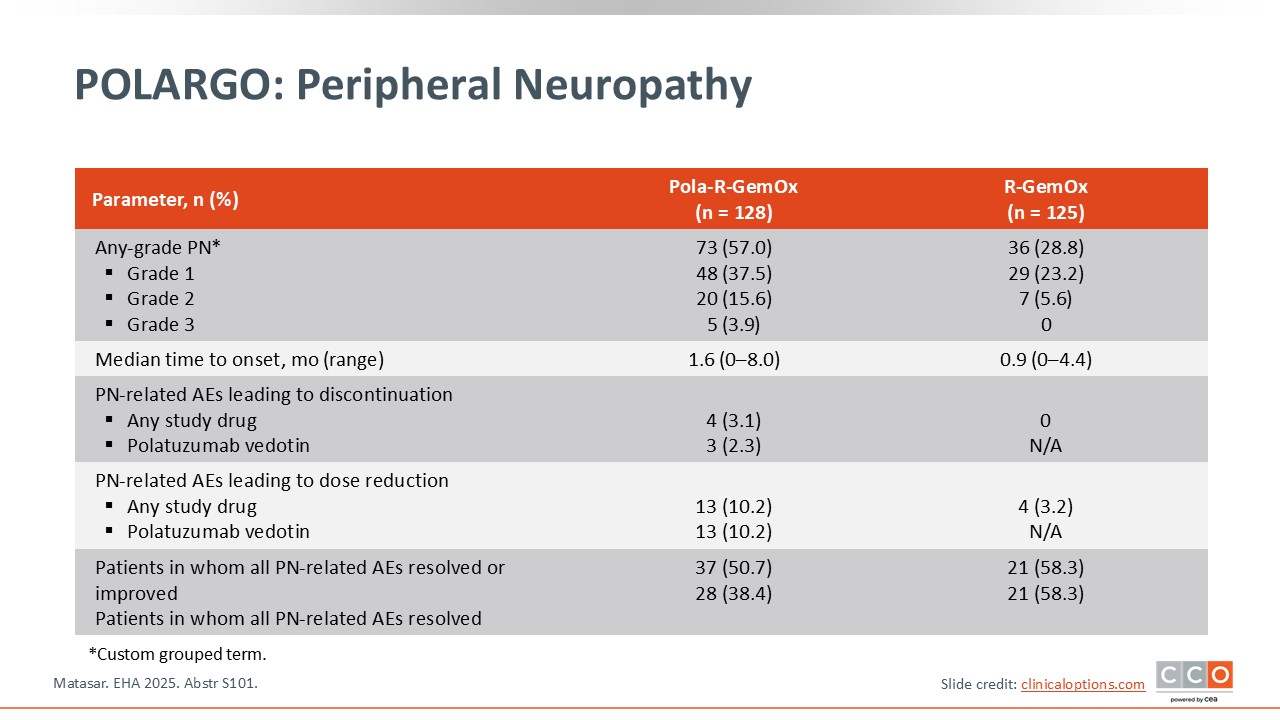
POLARGO: Peripheral Neuropathy
Pier Luigi Zinzani, MD, PhD:
One particular AE related to the use of polatuzumab vedotin that is important to be aware of is peripheral neuropathy. Grade 3 peripheral neuropathy was noted in approximately 4% of patients treated with Pola-R-GemOx vs no patients treated with R-GemOx. It is important to keep in mind that the AEs associated with polatuzumab vedotin are reversible in approximately 4-6 months.3
POLARGO: Investigator's Conclusions
Pier Luigi Zinzani, MD, PhD:
In conclusion, the addition of polatuzumab vedotin to R-GemOx improves outcomes, including a 40% reduction in relative risk of death vs conventional R-GemOx, in patients with transplant-ineligible R/R DLBCL. The survival benefit was consistent across all subgroups including regardless of the cell of origin. The safety profile of Pola-R-GemOx was consistent with its individual components, including infection and peripheral neuropathy. Overall, data from POLARGO supports Pola‑R‑GemOx as a treatment option in patients with R/R DLBCL that are considered transplant‑ineligible.3
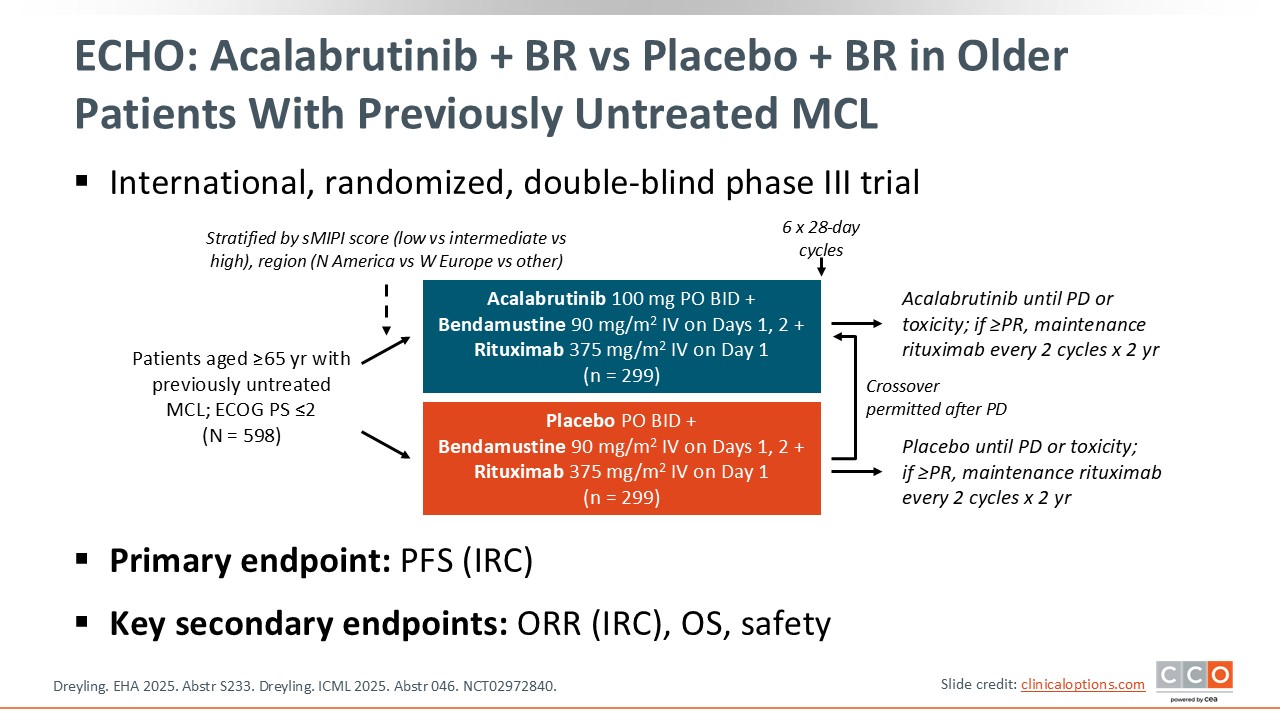
ECHO: Acalabrutinib + BR vs Placebo + BR in Older Patients With Previously Untreated MCL
Max S. Topp, MD:
The phase III ECHO trial is investigating acalabrutinib plus BR in patients with untreated MCL. In total, 598 patients aged 65 years or older were stratified by geographic region and simplified Mantle Call Lymphoma International Prognostic Index (MIPI) score. Patients received either acalabrutinib plus BR or placebo plus BR. Crossover was permitted after disease progression. The primary endpoint was PFS, and key secondary endpoints included the ORR, OS, and safety.
At EHA and ICML 2025, data from the high-risk population, defined as a high-risk MIPI, Ki-67 index ≥30%, presence of a TP53 mutation, and blastoid/pleomorphic histology, were presented.4
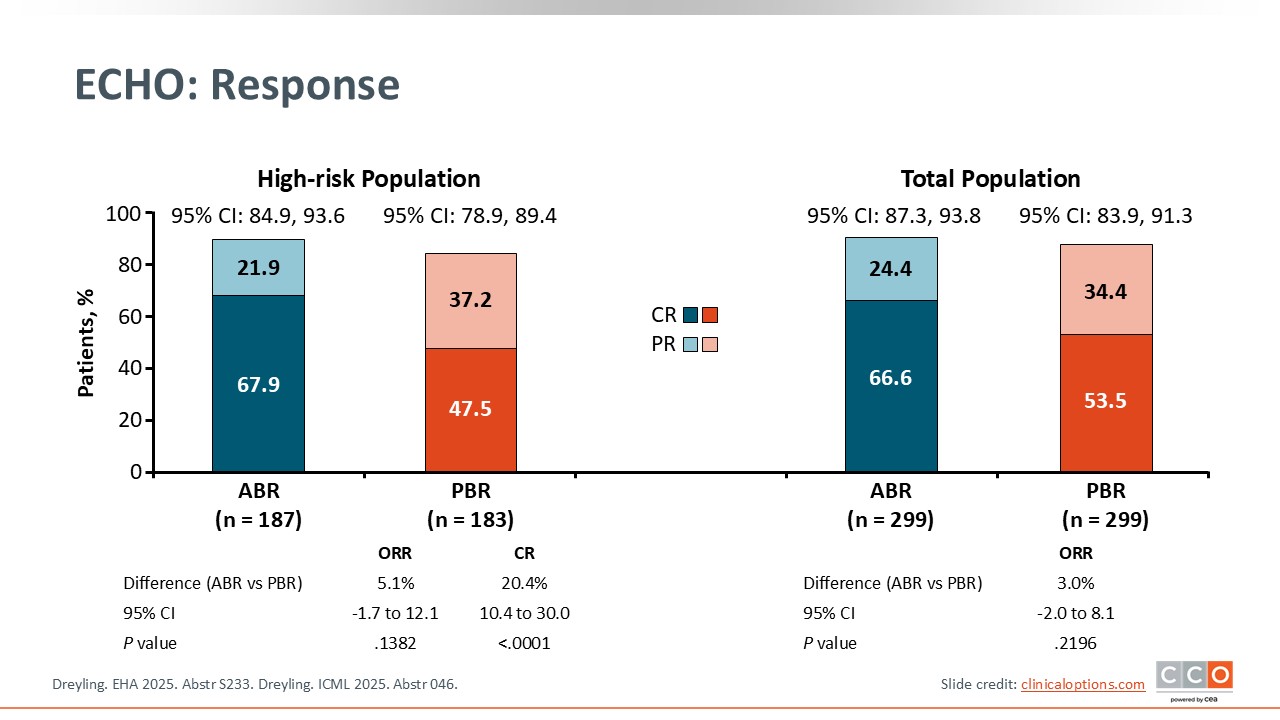
ECHO: Response
Max S. Topp, MD:
In the high-risk population, shown on the left, the ORR was 89.8% with acalabrutinib plus BR vs 84.7% with placebo plus BR. The best overall response of CR was observed in 67.9% of patients treated with acalabrutinib plus BR vs 47.5% who were treated with placebo plus BR (P <.0001).4
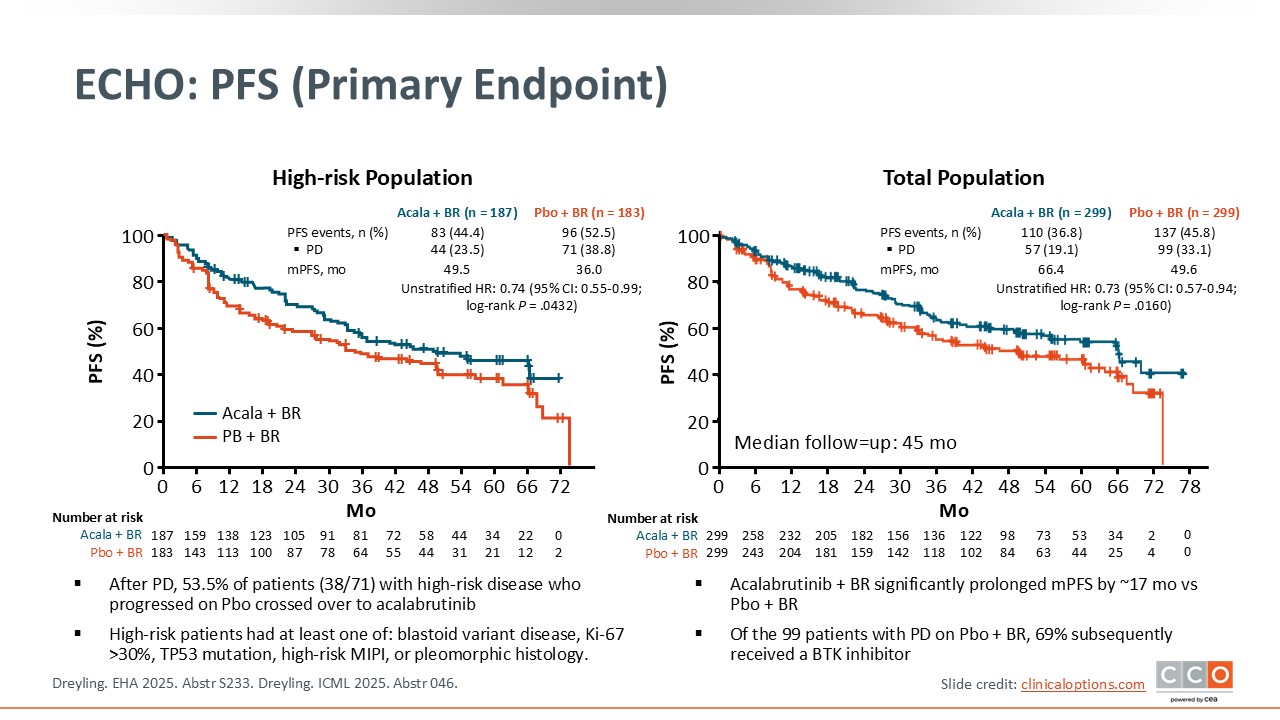
ECHO: PFS (Primary Endpoint)
Max S. Topp, MD:
In the high-risk patient population, the median PFS was 49.5 months for the acalabrutinib plus BR arm vs 36.0 months for the placebo plus BR arm, significantly prolonging survival by approximately 13.5 months (P = .0432). After disease progression, 38 patients with high-risk disease who initially received placebo crossed over to acalabrutinib.4
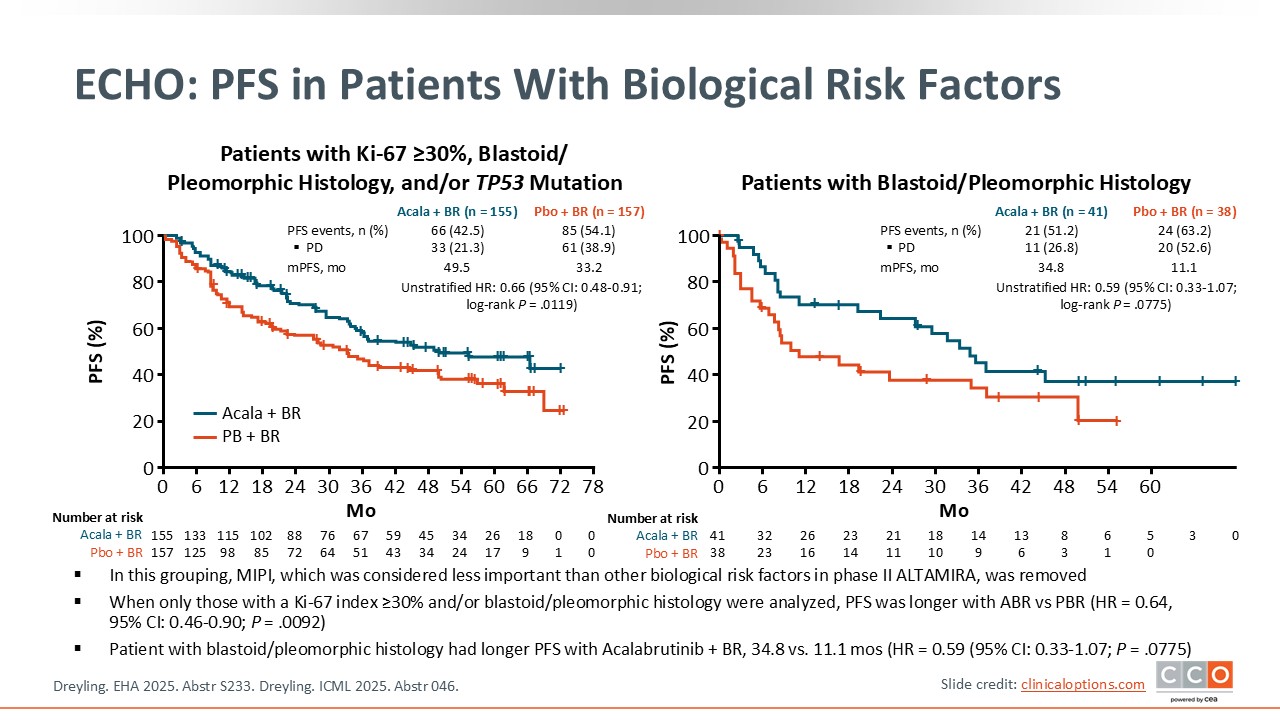
ECHO: PFS in Patients With Biological Risk Factors
Max S. Topp, MD:
The PFS benefit was observed across biologically high-risk subgroups. When considering only patients with a biological risk factor (ie, either a TP53 mutation, Ki-67 index ≥30%, and/or blastoid/pleomorphic histology), the addition of acalabrutinib to BR also demonstrated prolonged PFS vs placebo plus BR, median PFS was 49.5 vs 33.2 months (HR: 0.66; 95% CI: 0.48-0.91; P = .0119).4
Similarly, when only patients with a blastoid or pleomorphic histology were analyzed, PFS was significantly longer with acalabrutinib plus BR vs placebo plus BR (HR: 0.59; 95% CI: 0.33-1.07;
log-rank P = .0775), with a median PFS of 34.8 months vs 11.1 months, respectively, a difference of approximately 24 months.4
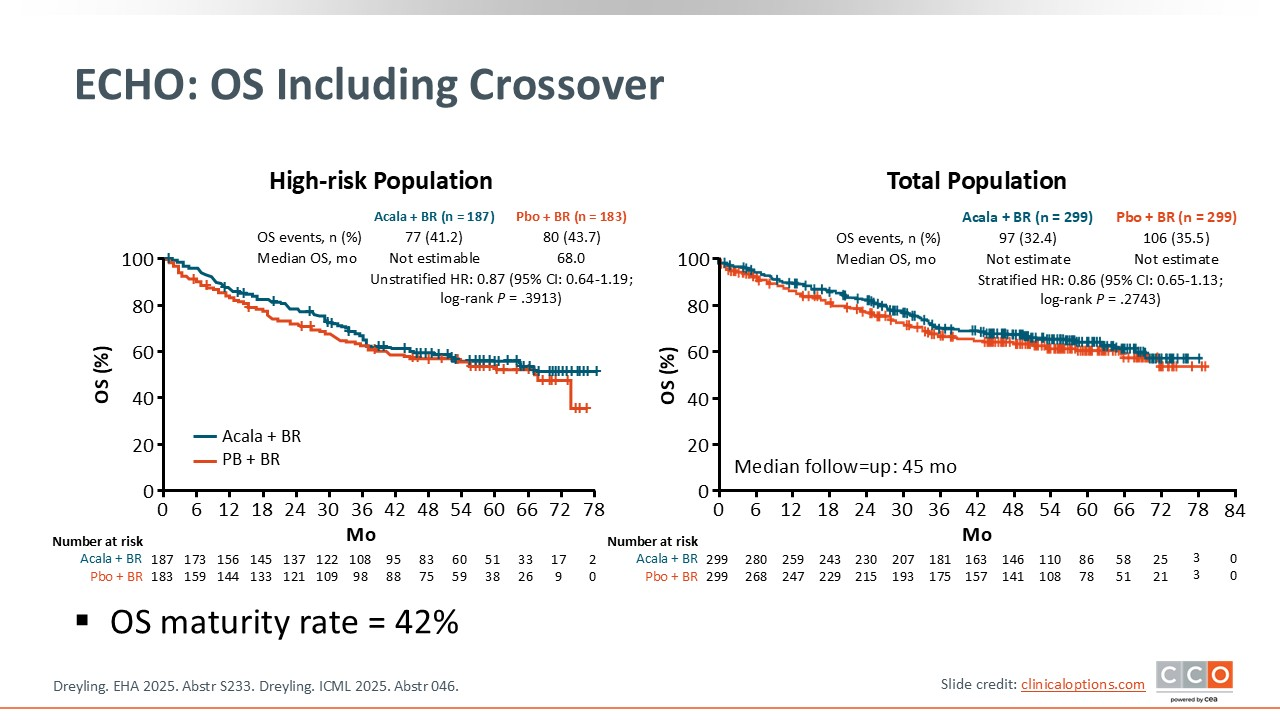
ECHO: OS Including Crossover
Max S. Topp, MD:
With an OS maturity rate of 42%, there was a positive trend in favor of acalabrutinib addition. When COVID-19–related deaths were censored, the trend was even more pronounced.4
ECHO: Investigator's Conclusions
Max S. Topp, MD:
Based on these data, consistent benefit across subgroups were seen with the addition of acalabrutinib to BR. Acalabrutinib plus BR improves PFS in older patients with untreated MCL, including those with high‑risk biology.4
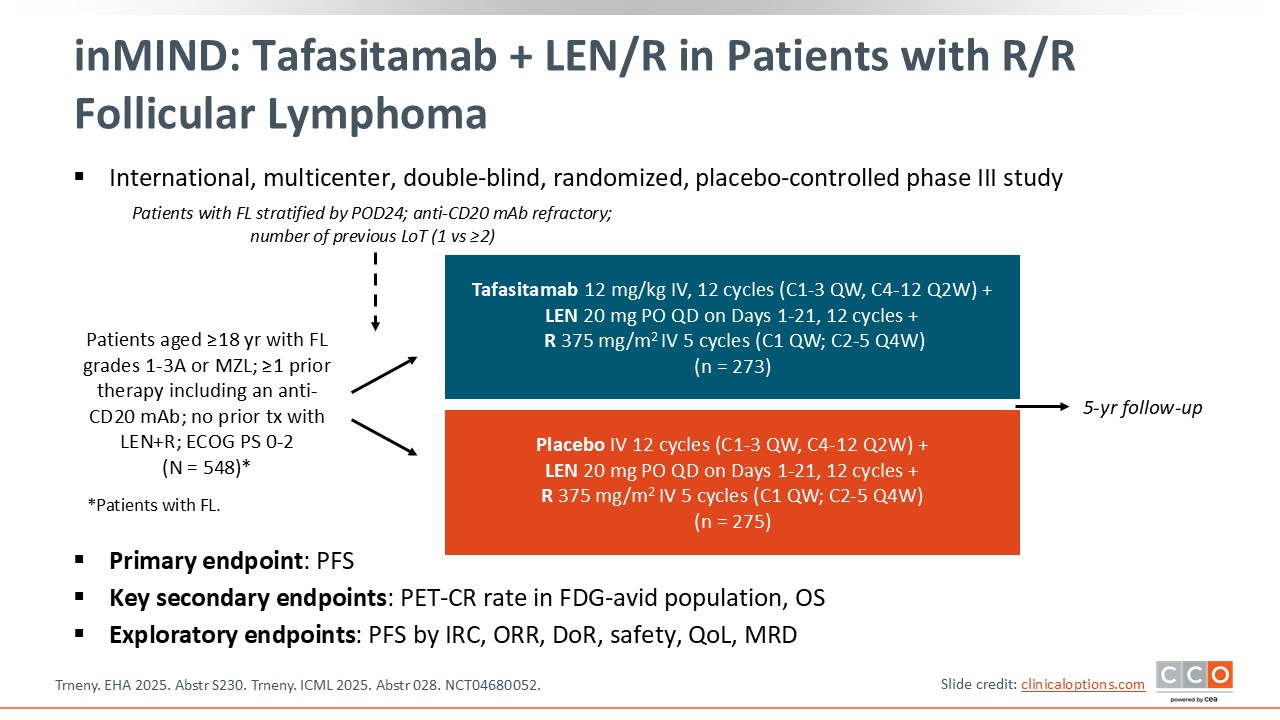
inMIND: Tafasitamab + LEN/R in Patients with R/R Follicular Lymphoma
Max S. Topp, MD:
inMIND is an international phase III trial that enrolled 548 patients with R/R follicular lymphoma (FL) or marginal zone lymphoma. Participants were randomized to receive either tafasitamab plus lenalidomide/rituximab or placebo plus lenalidomide/rituximab. The primary endpoint was PFS, and key secondary endpoints included the PET-CR rate in the FDG-avid population and OS.5
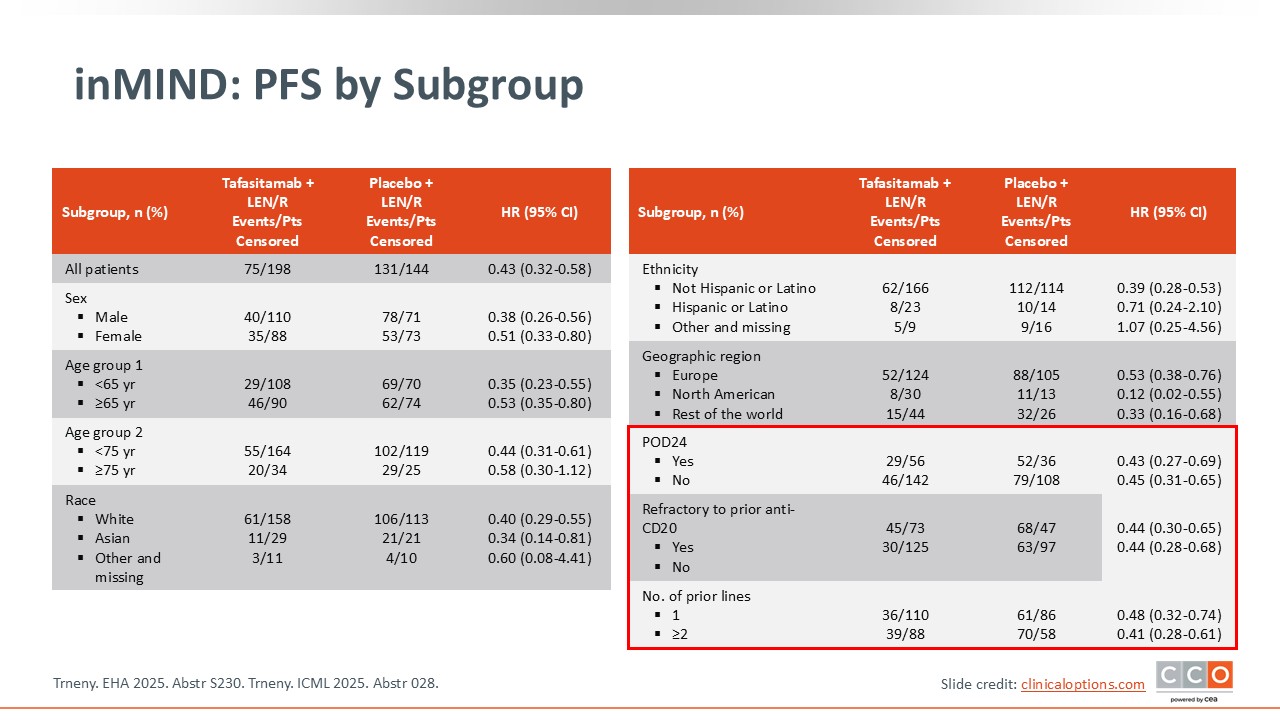
inMIND: PFS by Subgroup
Max S. Topp, MD:
The subgroup analysis showed that PFS benefit with the addition of tafasitamab did not depend on POD24 status, whether or not the patients were refractory to anti-CD20 antibody, or the number of prior lines of therapy. The addition of tafasitamab to lenalidomide/rituximab was superior to placebo plus lenalidomide/rituximab alone regardless of previous treatment. Overall, tafasitamab added to lenalidomide/rituximab resulted in a significant and clinically meaningful improvement in median PFS 22.4 months vs 13.9 months (HR: 0.43; P <.0001), representing a 57% reduction in risk of progression, relapse, or death.5
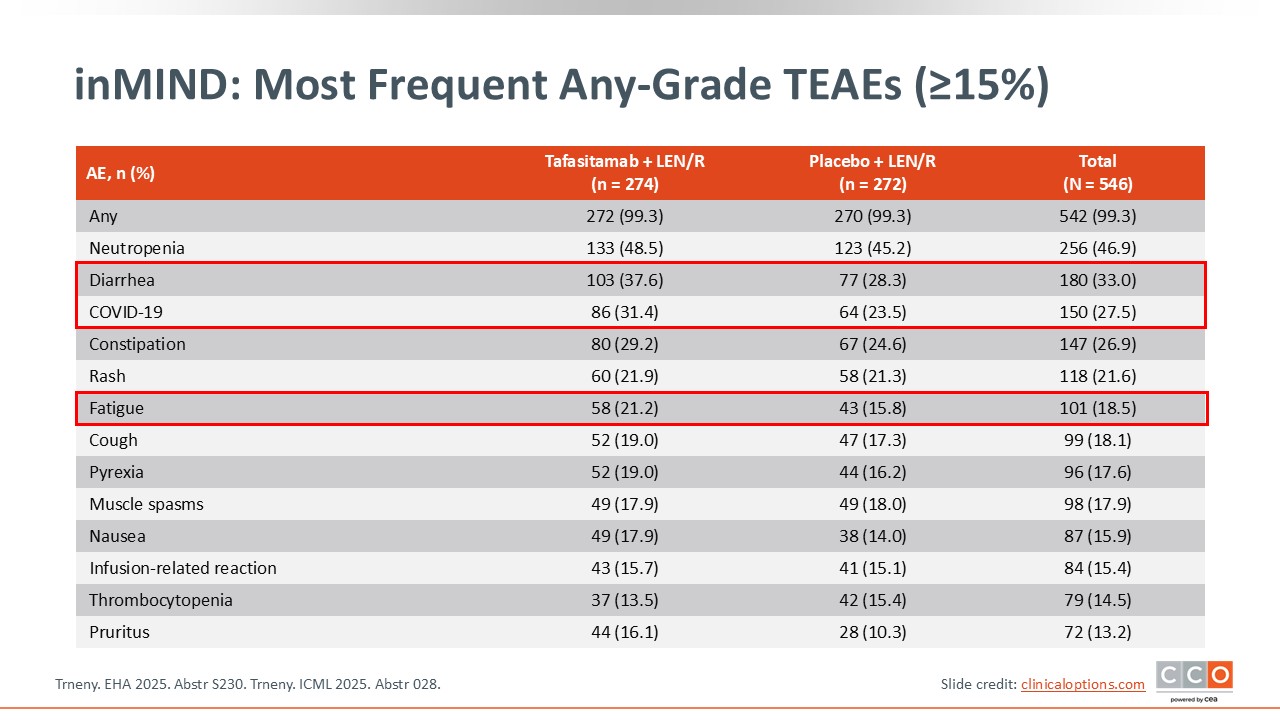
inMIND: Most Frequent Any-Grade TEAEs (≥15%)
Max S. Topp, MD:
When looking at safety, the rates of treatment-emergent AEs occurring in ≥15% of patients were the same between arms (99%). However, the rates of certain AEs, including any-grade diarrhea (37.6% vs 28.3%), COVID-19 (31.4% vs 23.5%), and fatigue (21.2% vs 15.8%), were higher in the cohort in which tafasitamab was added to lenalidomide/rituximab.5
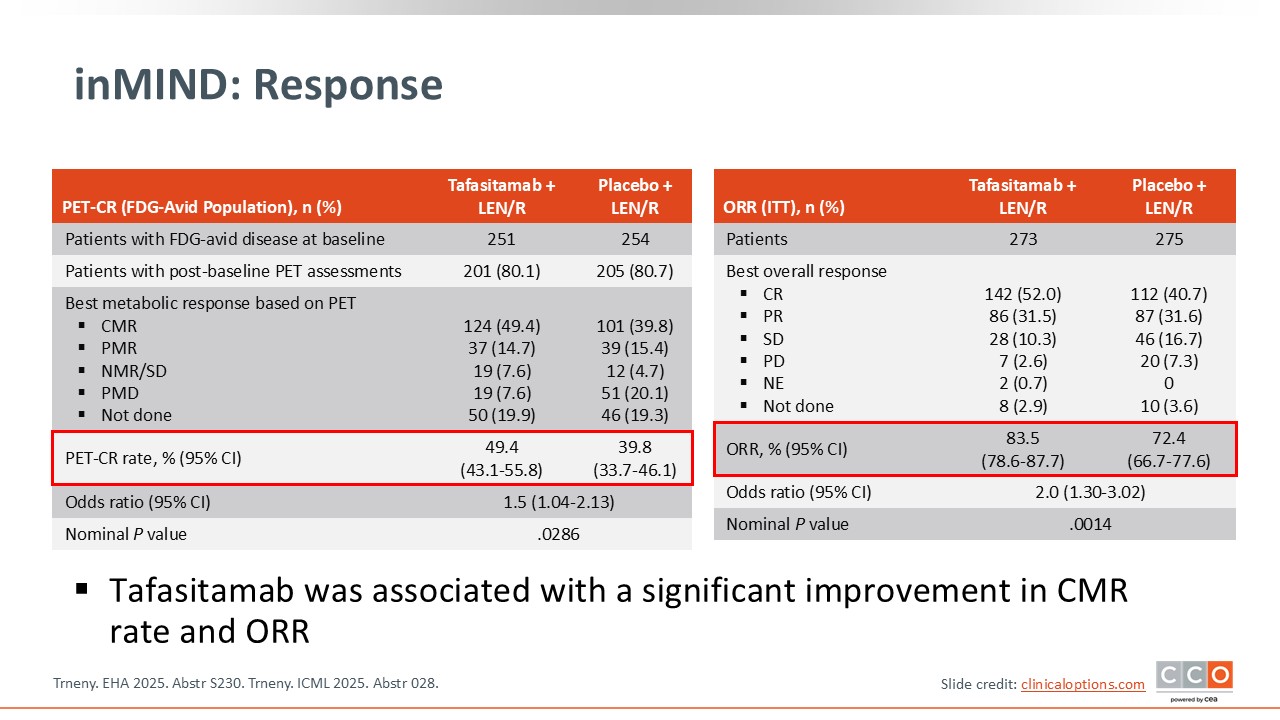
inMIND: Response
Max S. Topp, MD:
The PET-CR rate in the FDG-avid population was a key secondary endpoint. PET-CR was 49.4% with tafasitamab plus lenalidomide/rituximab vs 39.8% with placebo plus lenalidomide/rituximab. The ORR in the intention-to-treat population was another key endpoint, with ORR of 83.5% vs 72.4%.5
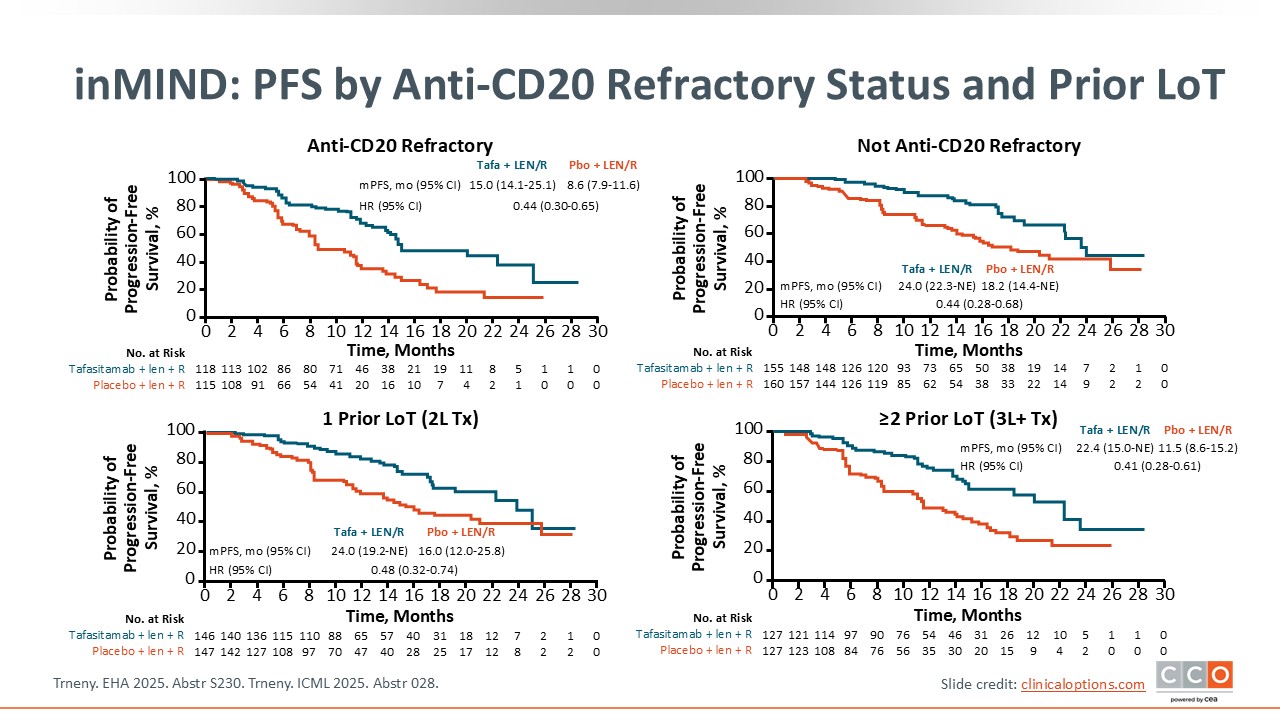
inMIND: PFS by Anti-CD20 Refractory Status and Prior LoT
Max S. Topp, MD:
PFS was analyzed by anti-CD20 refractory status and number of prior lines of therapy. The addition of tafasitamab consistently prolonged survival, with median PFS durations extending by nearly 11 months in the subgroup of patients who had received ≥2 prior lines of treatment.5
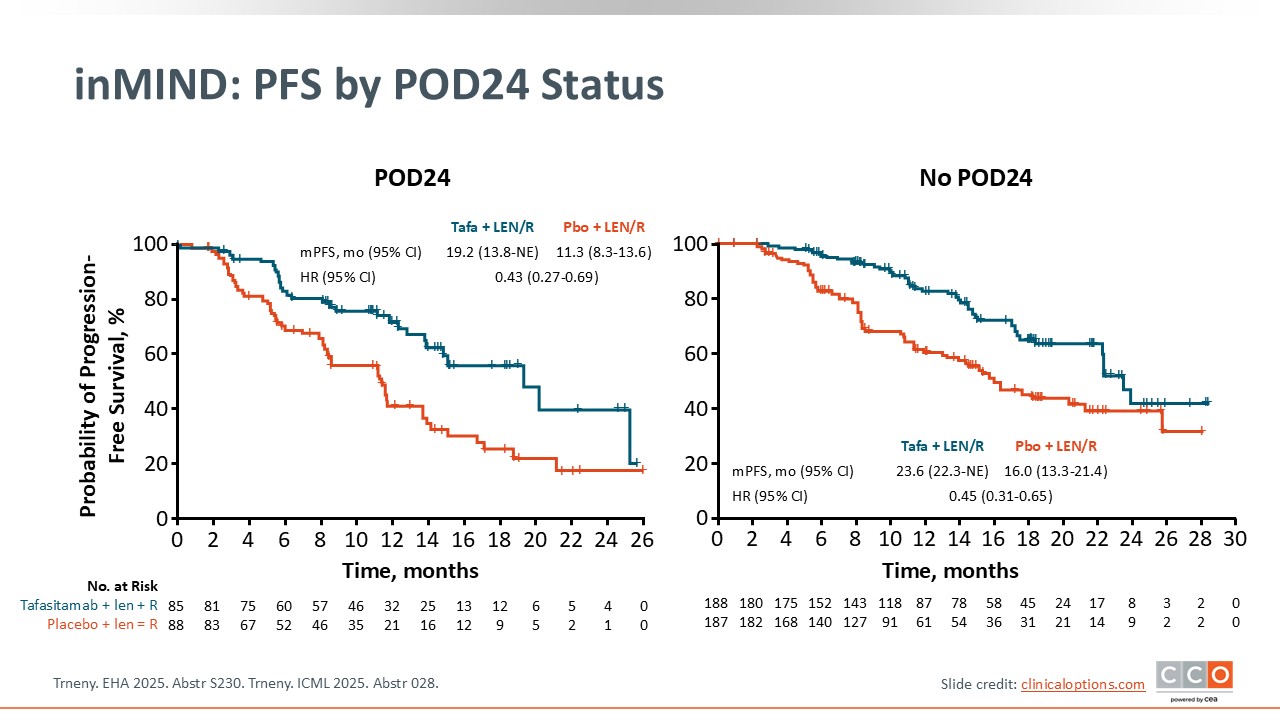
inMIND: PFS by POD24 Status
Max S. Topp, MD:
Similarly, when PFS was analyzed by POD24 status, tafasitamab continued to show an improvement in PFS. Median PFS was prolonged by almost 8 months with the addition of tafasitamab PFS benefit with the addition of tafasitamab did not depend on POD24 status, whether or not the patients were refractory to anti-CD20 antibody.5
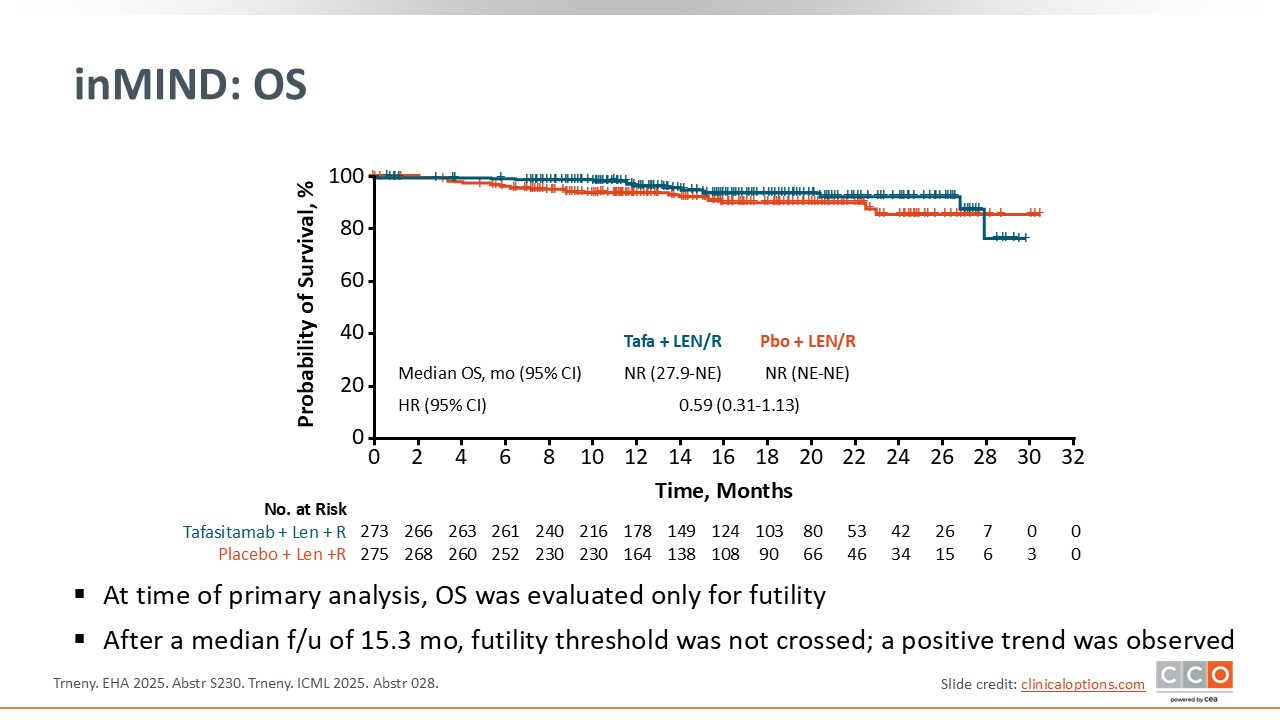
inMIND: OS
Max S. Topp, MD:
At the time of the primary analysis, OS was evaluated only for futility. However, although OS data are immature, with a median follow-up of 15.3 months, a trend favoring tafasitamab plus lenalidomide/rituximab compared with placebo plus lenalidomide/rituximab was observed.5
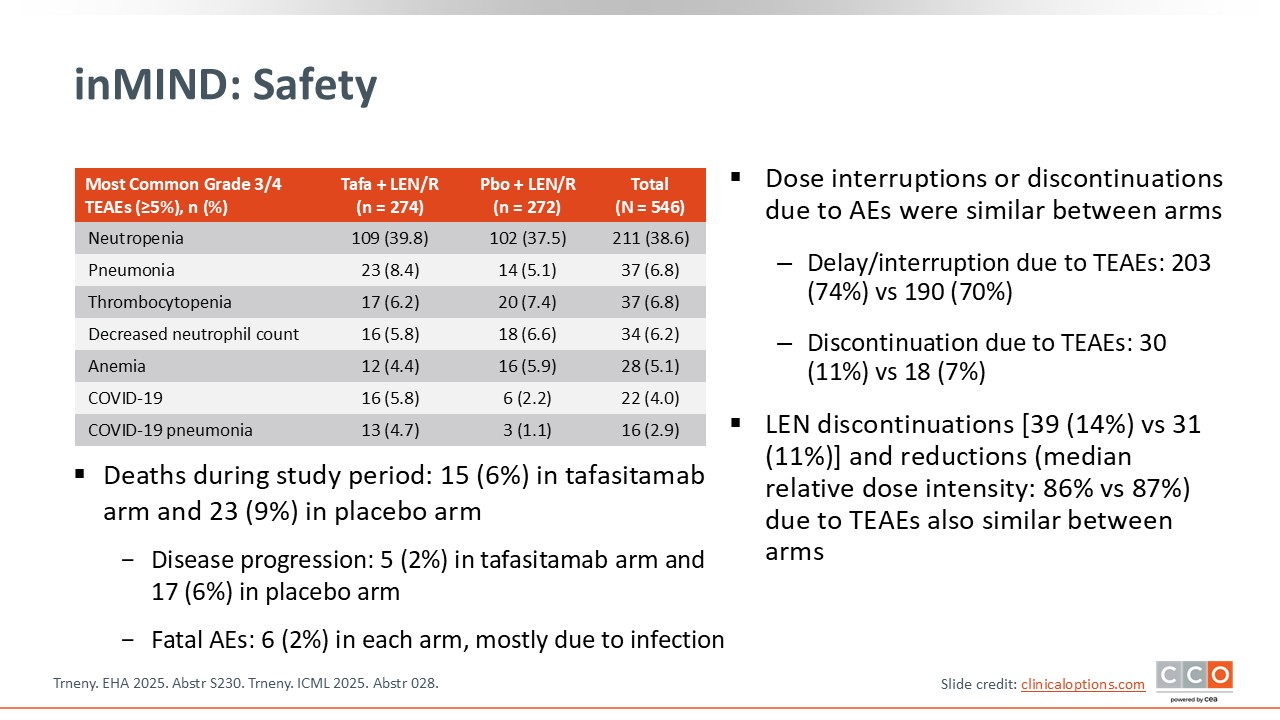
inMIND: Safety
Max S. Topp, MD:
The safety profile of tafasitamab plus lenalidomide/rituximab was manageable and consistent with expected AEs. The rates of dose interruptions or discontinuations were similar between arms. In total, 15 patients (6%) in the tafasitamab arm and 23 patients (9%) in the placebo arm died on study: 5 (2%) vs 17 (6%) because of disease progression and 6 (2%) in each arm because of fatal AEs.5
inMIND: Investigator's Conclusions
Max S. Topp, MD:
The addition of tafasitamab in this disease setting is very exciting. These data suggest that tafasitamab can be used as a chemotherapy‑free option in patients with R/R FL.5
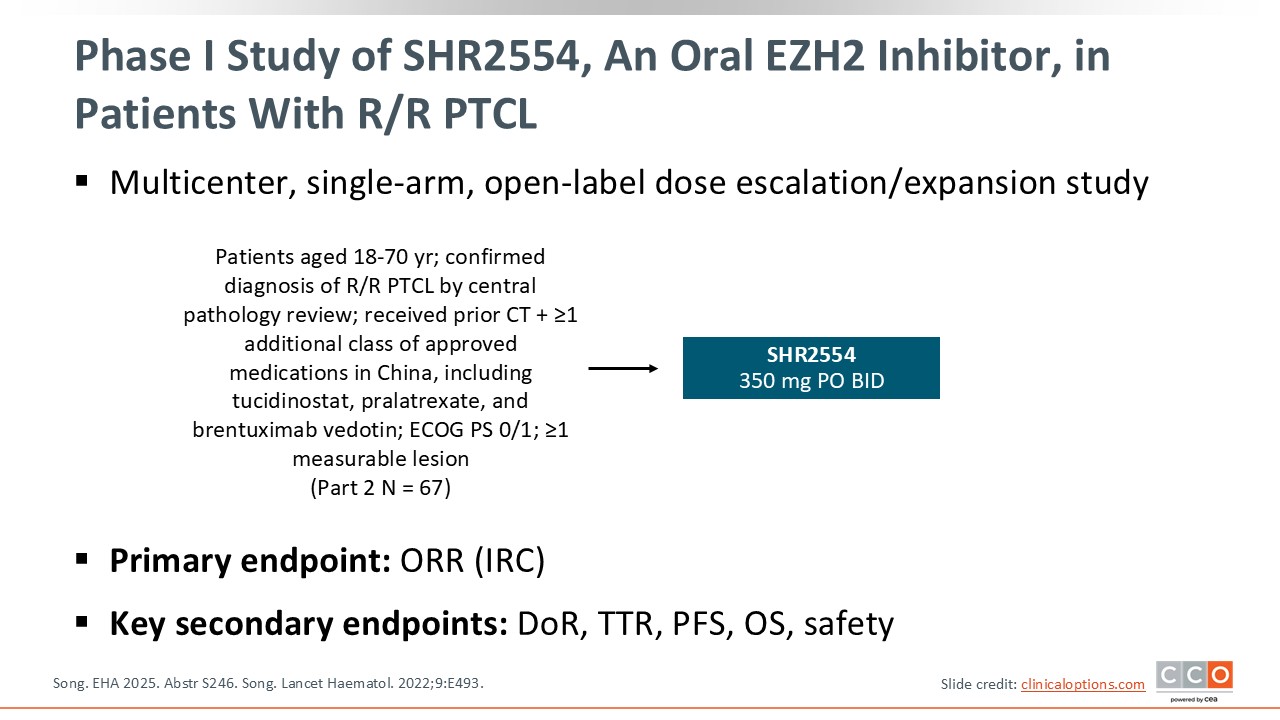
Phase I Study of SHR2554, an Oral Inhibitor Against EZH2, in Patients With R/R PTCL
Pier Luigi Zinzani, MD, PhD:
A pivotal phase I study is investigating a new oral EZH2 inhibitor, SHR2554, in patients with R/R peripheral T-cell lymphoma (PTCL). SHR2554 350 mg was administered orally twice daily (a dose determined to be the MTD in part 1). At EHA 2025, results from part 2 were presented. The trial enrolled patients aged 18-70 years who had received prior chemotherapy and at least 1 additional class of approved medications in China, including tucidinostat, an HDAC inhibitor; pralatrexate; or brentuximab vedotin. The primary endpoint was ORR by independent central review.6
Phase I Study of SHR2554: Baseline Characteristics
Pier Luigi Zinzani, MD, PhD:
The median patient age was 58 years. Several histologic subtypes were represented, including angioimmunoblastic TCL (AITL), PTCL-not otherwise specified, ALK-negative anaplastic lymphoma, ALK-positive anaplastic lymphoma, monomorphic epitheliotropic intestinal T-cell lymphoma, and others. Most patients were at advanced stages, and only 9% had bone marrow involvement before beginning treatment. The median number of prior lines of treatment was 2. Only 7.5% of patients had received hematopoietic stem cell transplantation.6
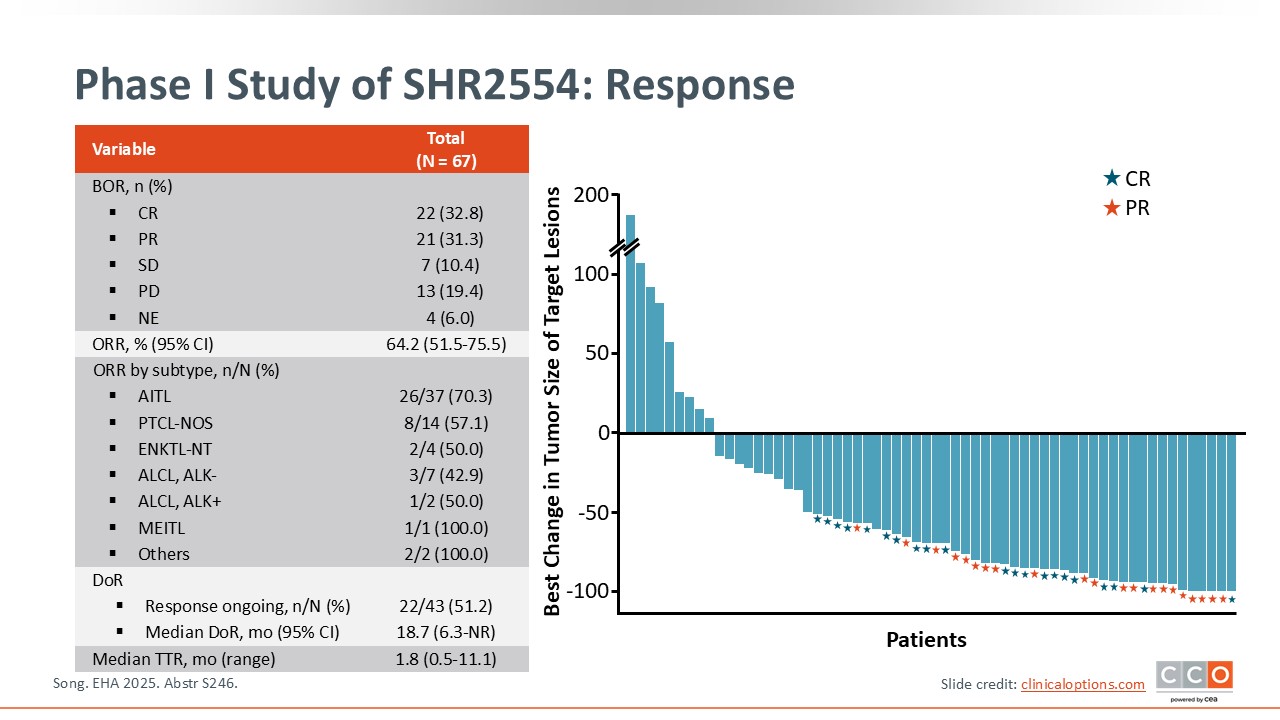
Phase I Study of SHR2554: Response
Pier Luigi Zinzani, MD, PhD:
In terms of tumor response, the ORR was 64.2%, and the complete metabolic response rate was 32.8%. These data are very impressive, especially when considering conventional treatments used in the United States or Europe, with much higher ORRs and CR rates, represented here in this waterfall plot.
In terms of ORRs according to subtype, in patients with AITL, the ORR was 70.3%. In those with PTCL-not otherwise specified, it was 57.1%.
Also important to note is that the median duration of response was more than 18 months, which is impressive, and the median time to response was less than 2 months.6
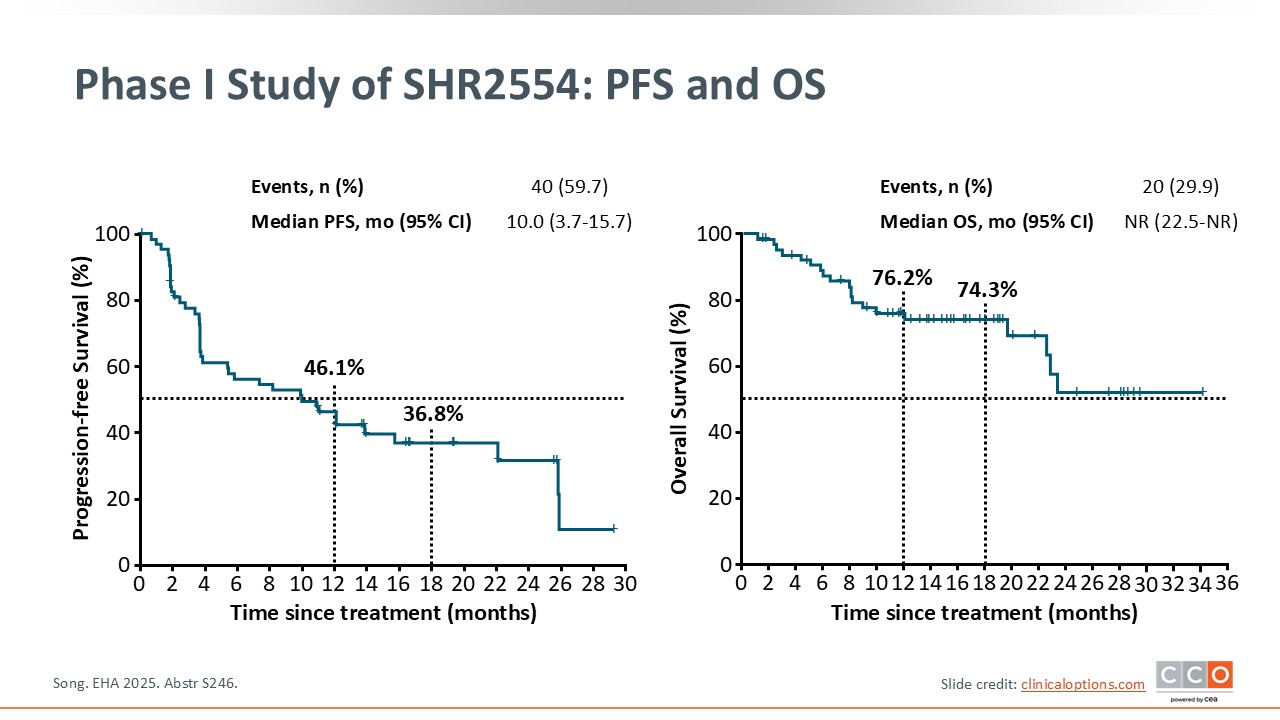
Phase I Study of SHR2554: PFS and OS
Pier Luigi Zinzani, MD, PhD:
PFS data are quite interesting. At 12 months, the PFS rate was 46.1%, and at 18 months, it was approximately 37%. The median PFS was 10 months.6
At 12 months, the OS rate was 76.2%, and at 18 months, it was 74.3%. The median OS was not reached.6
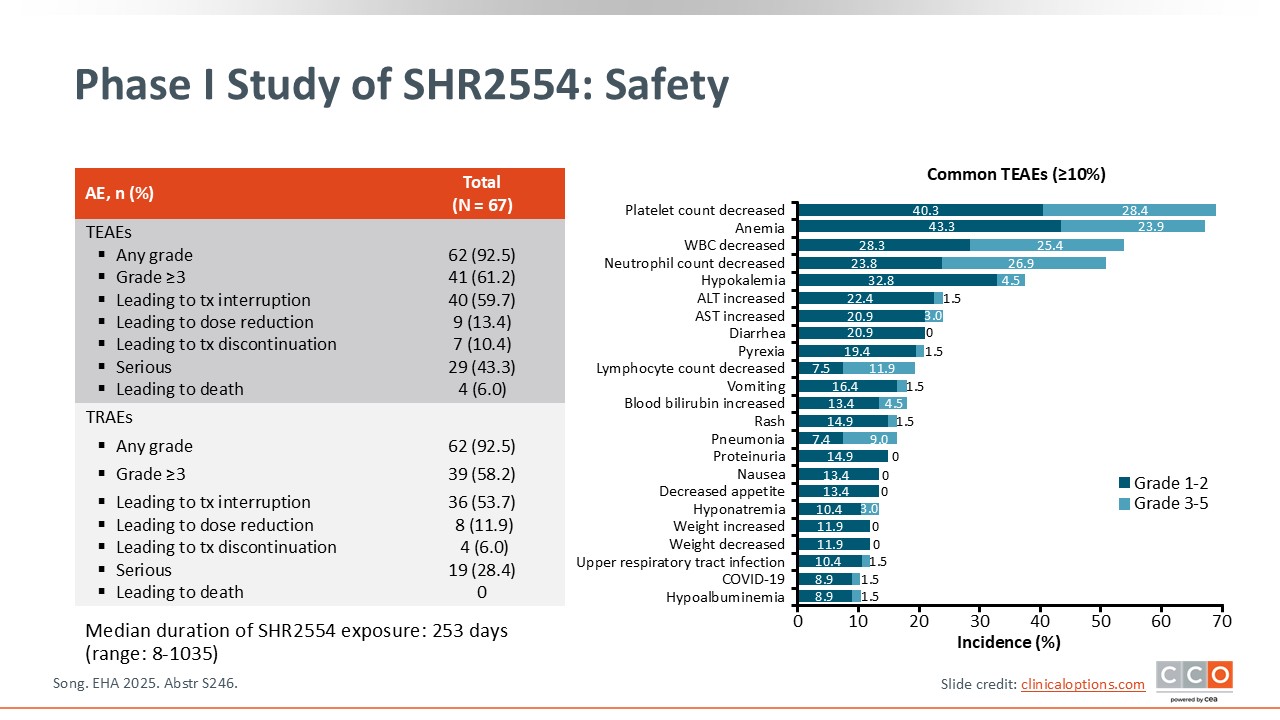
Phase I Study of SHR2554: Safety
Pier Luigi Zinzani, MD, PhD:
In terms of safety, some hematologic toxicities, represented by thrombocytopenia, neutropenia, and, in some patients, anemia, were reported. Some patients experienced an increase in alanine aminotransferase/aspartate aminotransferase levels, but most cases were grade 1/2 and were reversible within a few weeks.
Among the 67 patients who received SHR2554, no treatment-related deaths were reported.6
Phase I Study of SHR2554: Investigator's Conclusions
Pier Luigi Zinzani, MD, PhD:
These preliminary efficacy data on SHR2554, a new EZH2 inhibitor from China, are very impressive. An ORR >64% and a CR rate >30% are very encouraging. In addition, with a median duration of response of nearly 19 months, a median PFS of 10 months, and manageable toxicities, the future of our patients with R/R PTCL, regardless of histologic subtype, is hopeful.6