CME
HER2-Targeted ADCs in Genitourinary, Gastrointestinal, and Gynecological Malignancies: Safety and Adverse Event Management
Physicians: Maximum of 0.25 AMA PRA Category 1 Credit™
Released: September 03, 2025
Expiration: March 02, 2026
Activity
Introduction
In this activity, Zev A. Wainberg, MD; Alexandra Leary, MD, PhD; and Catherine Fahey, MD, PhD, explore optimal monitoring and management of adverse events (AEs) associated with HER2-targeted antibody–drug conjugates (ADCs) in the treatment of genitourinary (GU), gastrointestinal (GI), and gynecologic (GYN) malignancies. This session aims to bridge foundational information on HER2-targeted ADCs as potential treatment options in these settings with practical considerations for safety and AE management.
Please note that the slide thumbnails in this activity link to a PowerPoint slideset that also can be found here. The slideset also may be downloaded by clicking on any of the thumbnails within the activity.
Before continuing with this educational activity, please take a moment to answer the following questions.
HER2-Targeted ADC Design, Mechanism of Action, and Implications for Adverse Events
The development of HER2-targeted therapies has dramatically changed the treatment landscape for many patients with cancer. Initially, HER2-targeted antibodies demonstrated improved outcomes in patients with breast cancer and HER2 amplification. More recently, novel HER2-targeted strategies using ADCs have also expanded into other solid tumor types, including gastric and gastroesophageal junction (GEJ), colorectal, bladder, biliary tract, and endometrial cancers.
In this module, we focus on HER2-targeted ADCs, as this treatment class has been shown to have the most significant efficacy in various solid tumors.
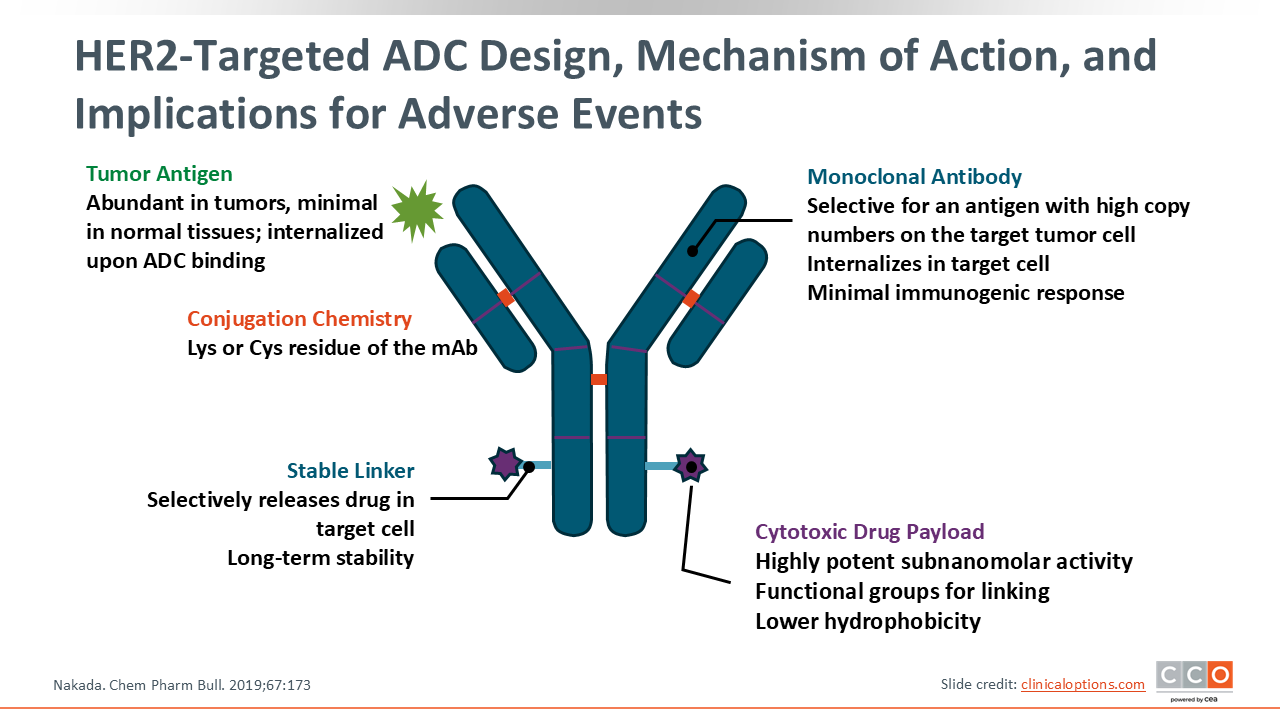
ADCs deliver a cytotoxic payload to antigen‑expressing tumor cells via a monoclonal antibody, essentially functioning as a targeted chemotherapy. Following binding to the HER2 receptor on a cancer cell, the ADC–receptor complex is internalized into the cell. Within the lysosome, the linker is cleaved, releasing the cytotoxic payload. This release mechanism not only kills the target cell but can also induce a “bystander effect,” where the payload diffuses out to kill adjacent tumor cells that may not express the target antigen, while largely sparing other healthy tissues throughout the body. Because ADCs target specific cells rather than systemic infusion of cytotoxic chemotherapy, there are fewer observed AEs—however, as with any cancer therapy, AEs do still occur. The AE profile for each ADC can depend on both the monoclonal antibody and the cytotoxic payload, so each ADC can have a distinct AE profile that should be considered.
There are currently 3 different HER2 ADCs that are approved worldwide, including trastuzumab emtansine (T-DM1) for HER2-positive breast cancer; T-DXd for HER2-positive, HER2-low, and HER2-ultralow breast cancer, HER2-positive gastric or GEJ adenocarcinoma, HER2-positive solid tumors, and HER2-mutated non-small-cell lung cancer; and DV, which is approved in China for HER2-positive gastric cancer, HER2-positive urothelial cancer, and HER2-positive breast cancer.
In this module, we discuss current AE monitoring and management for the 2 ADCs approved for patients with GU, GI, and GYN cancer—T-DXd and DV.
Trastuzumab Deruxtecan: HER2-Targeted ADC
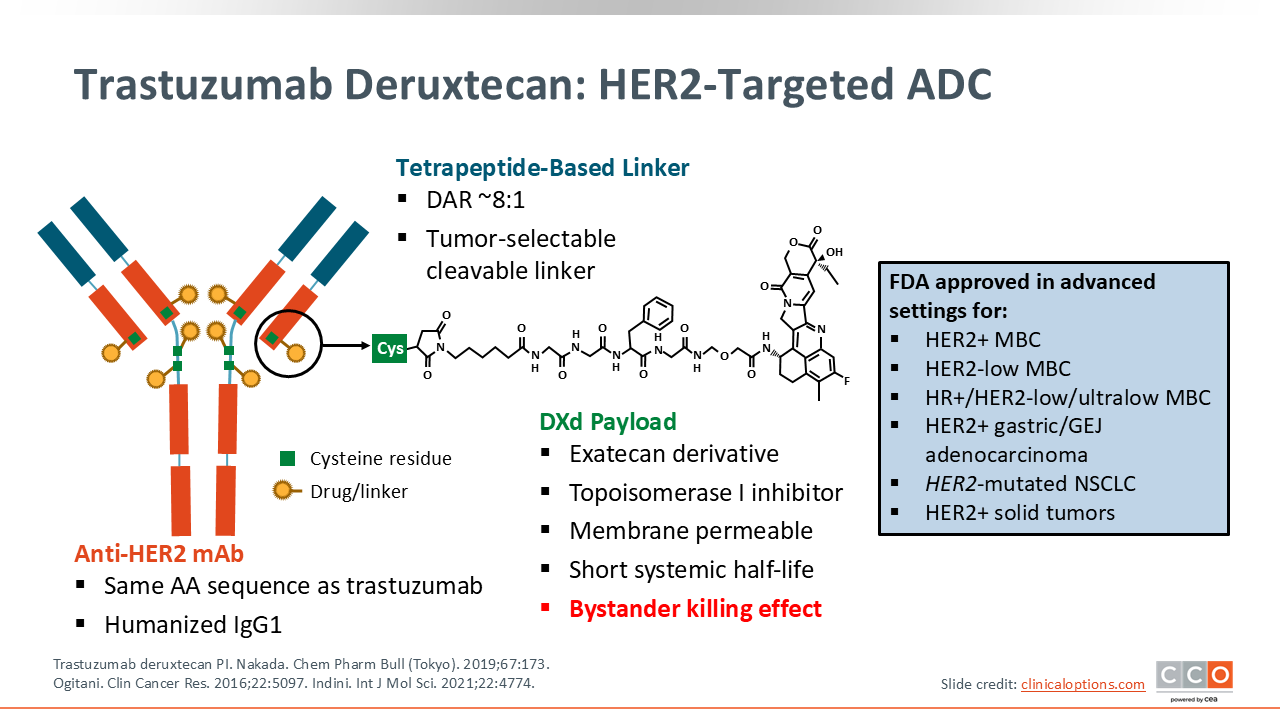
In April 2024, the FDA granted the first tumor‑agnostic accelerated approval for a HER2‑directed ADC: T-DXd, which is now approved for adults with previously treated, unresectable or metastatic, HER2‑positive (IHC 3+) solid tumors.1 This approval allows use across any solid tumor with high HER2 expression when no satisfactory alternatives exist.2
T-DXd is an ADC with a HER2-targeted monoclonal antibody linked to a deruxtecan payload in an 8:1 drug-to-antibody ratio with a tumor-selective cleavable linker. The deruxtecan payload is a topoisomerase I inhibitor that is membrane permeable, with a short systemic half-life. Based on these structural characteristics, T-DXd has a unique profile with common AEs known to be associated with traditional chemotherapy, such as fatigue, nausea, and cytopenias, along with some AEs that may be less familiar to some health care professionals, including ILD.
DESTINY-PanTumor02: Safety Summary With T-DXd
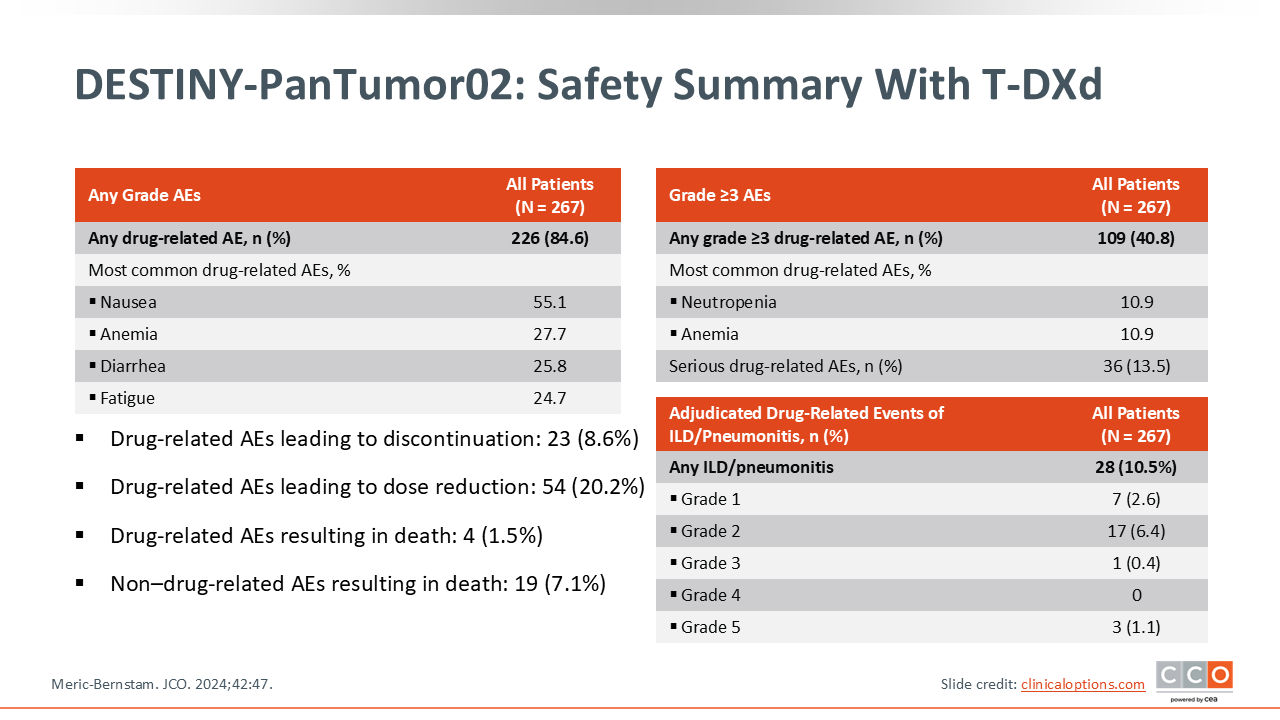
The DESTINY-PanTumor02 is an open-label, multicenter, phase II trial that assessed the efficacy and safety of T-DXd at a dose of 5.4 mg/kg once every 3 weeks in patients with locally advanced, unresectable, or metastatic solid tumors and HER2 expression (IHC 3+ or 2+)—including endometrial, cervical, ovarian, bladder, biliary tract, and pancreatic cancers or those with other solid tumors excluding breast, colorectal, gastric, and non-small-cell lung cancers (NSCLC)—with disease progression after 1 or more prior treatment or with no satisfactory alternatives. The results of this trial formed the basis for the FDA tumor agnostic approval of T-DXd for adults with previously treated, unresectable or metastatic, HER2 positive (IHC 3+) solid tumors.2
In this trial, 84.6% experienced a drug-related treatment-emergent adverse event (AE) (84.6%) and 40.8% had a grade ≥3 event. The most common AEs with T-DXd included nausea, anemia, diarrhea, and fatigue, and the most common grade ≥3 AEs included neutropenia and anemia.
As mentioned, ILD/pneumonitis is an established risk with T-DXd and this AE was observed in 28 patients (10.5%). Most incidence of ILD/pneumonitis were low-grade events, but there were 3 deaths (1.1%) related to ILD/pneumonitis.
In addition to ILD, a small number of patients may experience cardiac AEs while receiving T-DXd. In a previous presentation of this trial, 2.6% of patients were reported to have experienced ejection fraction decrease and 1 patient experienced cardiac failure.3
Managing ILD/Pneumonitis With T-DXd
ILD/pneumonitis has been associated with various ADCs, including T‑DXd. ILD refers to a group of disorders that cause inflammation and/or scarring (fibrosis) of the lung interstitium and alveoli, impairing gas exchange and typically presenting with exertional dyspnea and a dry cough.4
In DESTINY‑PanTumor02, ILD/pneumonitis occurred in 10.5% of patients, most (9.0%) were grade 1/2 and there were 3 grade 5 (1.1%) fatalities.2
Based on these data, the US prescribing information carries a boxed warning for ILD/pneumonitis. HCPs should educate patients; monitor for cough, dyspnea, fever or hypoxia; and promptly evaluate suspected cases with imaging and infection workup. HCPs should also monitor for signs and symptoms of ILD during treatment.
If ILD is suspected, T-DXd should be held and the patient should undergo a full workup, including radiographic imaging and blood culture, CBC, or other blood tests as needed to either rule out or confirm ILD. Also consider pulmonologist consult and, if indicated, infectious diseases consult for patients experiencing pulmonary symptoms.
For confirmed grade 1 ILD events, continue to hold T-DXd and consider starting corticosteroids. If grade 1 ILD resolves in ≤28 days, T-DXd may resume at the same dose or if resolution takes >28 days, T-DXd can be resumed at 1 dose level lower and do not reescalate.5,6 For symptomatic (grade ≥2) ILD/pneumonitis, promptly initiate a course of systemic corticosteroids and permanently discontinue T-DXd.
The starting dose for T-DXd is 5.4 mg/kg for IHC 3+ solid tumors or 6.4 mg/kg for HER2-positive gastric/GEJ cancer. If dose reduction is needed, step down to 4.4 mg/kg at first reduction and then 3.2 mg/kg at second reduction from 5.4-mg/kg starting dose or 5.4 mg/kg and then 4.4 mg/kg for 6.4-mg/kg starting dose and do not reescalate. If a further reduction would be required after the second dose reduction, T-DXd should be discontinued. If a dose is missed, administer it as soon as possible and continue on the every-3-week schedule.6
Pooled analyses across modern trials show that adjudicated ILD occurs in approximately 10% to 15% of patients, with most events being low grade.7
Detecting and Managing T-DXd–Related Interstitial Lung Disease: The 5 “S” Rules
The 5 “S” rules for T-DXd–related ILD can help guide HCPs considerations for potential ILD in patients receiving T-DXd.
Screening: Carefully assess patients before starting therapy. Careful patient selection is necessary prior to starting T-DXd to optimize the monitoring strategies according to baseline risk. Ongoing screening is continued throughout treatment with regular clinical evaluations to detect any signs/symptoms of ILD.
Scan: Radiologic imaging, particularly high-resolution chest CT scans, remains the primary diagnostic method for ILD. A baseline scan is recommended, followed by repeat imaging every 6-12 weeks during treatment.
Synergy: Reducing the risk of ILD requires teamwork, involving patient education, training of the entire care team, and multidisciplinary management once ILD is suspected. Use a coordinated team—patient/caregiver, oncology (including infusion nurses), clinic staff, plus pulmonology and infectious diseases—to detect and manage AEs.
Suspending treatment: Stop T-DXd immediately if ILD is suspected and resume only after full resolution from a grade 1 ILD event. For grade ≥2 ILD events, permanently discontinue T-DXd.
Steroids: Corticosteroids remain the primary treatment for T-DXd–induced ILD, with the dosage according to the severity of toxicity. Consider corticosteroids for grade 1 ILD, but always begin corticosteroids for any symptomatic (grade ≥2) ILD event.
Managing Nausea/Vomiting in Patients on T-DXd
T-DXd is highly emetogenic, and nausea/vomiting can be delayed after the start of treatment. Prophylactic antiemetics are recommended with each infusion, following local guidelines for prevention of chemotherapy-induced nausea and vomiting.6
The NCCN antiemesis guidelines recommend managing high-emetic-risk regimens with Day 1, 4-drug prophylaxis (NK1 receptor antagonist, 5-HT3 receptor antagonist, dexamethasone, and olanzapine) and continue antiemetic coverage on Days 2-4.8
Counsel patients that symptoms may start or worsen after Day 1, schedule prophylaxis for first 4 days after treatment, and provide take-home antiemetics.6
T-DXd: Strategies to Prevent and Manage Neutropenia and Thrombocytopenia Associated With T-DXd Treatment
Cytopenias, including neutropenia and thrombocytopenia, are key AEs associated with ADCs like T-DXd. Management principles are similar to other anticancer therapies, and the prescribing information instructs HCPs to monitor CBC with differential before treatment, prior to each dose, and as clinically indicated, managing through treatment interruption and/or dose reduction. Most cytopenia events are low grade with a low risk of febrile neutropenia. Median time to onset of neutropenia is 2-3 weeks after starting treatment, and the median onset for thrombocytopenia is approximately 4 weeks.
Hold T-DXd for grade 3 neutropenia until recovery to grade ≤2, then resume at the same dose. For grade 4 or febrile neutropenia, interrupt until recovery to grade ≤2 and reduce by one dose level.
Hold T-DXd for grade 3 thrombocytopenia until recovery to grade ≤1 and then maintain dose. For grade 4 thrombocytopenia, interrupt until recovery to grade ≤1 and reduce by 1 dose level.
Monitoring and Management of LVEF With T-DXd
Decreased left LVEF with T-DXd is an uncommon but serious AE associated with many HER2-targeted therapies, including T-DXd. LVEF and cardiac function should be assessed before starting T-DXd and at regular intervals during treatment as clinically indicated. In pooled analyses of the 5.4-mg/kg dose population, LVEF decrease was reported in 4.6%; in gastric cancer at 6.4 mg/kg, 8% had asymptomatic grade 2 decreases on echocardiography.6
In practice, many centers assess LVEF approximately every 3 months in the early phase of treatment and may extend the monitoring interval for stable patients.
Management is by LVEF value and changes from baseline. For patients with LVEF >45% with a 10% to 20% absolute decrease, continue T-DXd treatment with consistent LVEF monitoring. For LVEF of 40% to 45% with <10% decrease from baseline, continue T-DXd treatment and repeat LVEF monitoring within 3 weeks. For LVEF of 40% to 45% with a 10% to 20% decrease, interrupt T-DXd therapy and repeat LVEF assessment within 3 weeks. If LVEF is not recovered to within 10% of baseline, permanently discontinue T-DXd, but if LVEF is recovered, resume T-DXd at the same dose. For LVEF <40% or >20% absolute decrease, interrupt T-DXd and repeat LVEF assessment within 3 weeks and, if confirmed, permanently discontinue T-DXd. For any patients with symptomatic congestive heart failure, permanently discontinue T-DXd.
Disitamab Vedotin: HER2-Targeted ADC Mechanism and Current Indications
DV is a HER2‑directed ADC that delivers the microtubule inhibitor monomethyl auristatin E (MMAE) via a protease‑cleavable linker, concentrating cytotoxic effect in HER2‑expressing tumors. In China, the National Medical Products Administration (NMPA) has approved DV for HER2‑overexpressing (IHC 2+ or 3+) locally advanced/metastatic gastric/GEJ adenocarcinoma after ≥2 previous lines of chemotherapy and in HER2‑overexpressing urothelial carcinoma after previous platinum-based chemotherapy. In May 2025, the NMPA also approved DV monotherapy for HER2‑positive metastatic breast cancer with liver metastases after previous treatment with trastuzumab and taxanes.9
Disitamab Vedotin: Safety Summary in Patients With HER2+ UC
A combined analysis of 2 open-label, multicenter, single-arm phase II clinical trials evaluated the efficacy and safety of DV in patients with HER2-positive (IHC 3+ or 2+) locally advanced or metastatic urothelial cancer. Patients with bladder cancer who had progression after ≥1 prior lines of systemic chemotherapy were enrolled and received DV 2 mg/kg intravenously every 2 weeks.10
In this trial, all patients experienced a treatment-related AE and 54.2% had a grade ≥3 event. The most common AEs with DV included peripheral neuropathy, leukopenia, aspartate aminotransferase increase, and neutropenia, and the most common grade ≥3 AEs included peripheral neuropathy, neutropenia, and asthenia.
Drug-related AEs leading to discontinuation were reported in 19 patients, and drug-related AEs leading to dose reduction occurred in 32 patients, with only 1 of these needing to discontinue DV. No grade 5 treatment-related AEs were observed.
Key Toxicities With DV and How to Monitor
Based on the AEs reported in clinical trials, dose modifications for DV focus on patients who experience myelosuppression, peripheral sensory symptoms/neuropathy, and transaminase elevations. For practical monitoring for patients while receiving DV, check CBC and liver function tests prior to each dose and as clinically indicated; assess for new paresthesias/numbness each cycle; and review concomitant drugs for CYP3A4 interactions (MMAE is a CYP3A4 substrate, so strong inhibitors/inducers may alter exposure).9
Disitamab Vedotin and Hematologic Events
In published clinical trials, when hematologic events occur, dose holds and/or dose reductions were used to manage these AEs.10 For grade 1/2 cytopenias, DV was continued at the original dose (2.0 mg/kg). For grade 3 hematologic AEs, DV should be held at the first occurrence until recovery to grade ≤1 and then resumed at the same dose. For a second or third grade 3 occurrence, DV should be resumed 1 dose level lower (1.5 mg/kg), and similarly, for a third recurrence, DV should be resumed at 1.0 mg/kg. However, for a fourth recurrence, DV should be discontinued (or continued only if the HCP judges that the benefit outweighs risk). For grade 4 hematologic AEs, hold DV at first occurrence and then resume 1 dose level lower, but at second recurrence, DV should be discontinued (or continue only if the HCP judges that the benefit outweighs risk).
If a drug‑related grade ≥3 hematologic AE occurs, it is recommended to perform twice‑weekly laboratory tests and if not recovered to a level permitting treatment within 28 days of the planned dose date, stop DV.9,10
Disitamab Vedotin and Transaminase Elevations
Hepatotoxicity was another common AE in DV clinical trials. For grade 1/2 hepatotoxicity (alanine aminotransferase/aspartate aminotransferase increase), DV treatment can be continued. For grade 3 events, at first occurrence DV should be held and then resumed 1 level lower once recovered. For a second grade 3 event, DV should be held and then resume 2 levels lower than the original dose. At a third grade 3 event recurrence, DV should be discontinued (with an exception to continuation treatment per HCP judgment). Any grade 4 hepatotoxicity event warrants DV discontinuation.
If a drug‑related grade ≥3 hepatotoxicity event occurs, obtain twice‑weekly blood chemistry tests to monitor for improvement. If not recovered to grade ≤2 (or baseline) within 28 days, permanently discontinue DV.9,10
Disitamab Vedotin and Peripheral Sensory Neuropathy
In clinical trials, peripheral sensory symptoms/neuropathy was a common AE and grade 3 events also occurred in almost 20% of patients. When peripheral sensory symptoms/neuropathy occurs in patients who are receiving DV, treatment should be paused quickly and the dose reduced on recurrence. Symptoms should be managed with supportive care treatment as necessary. For grade 1 symptoms, DV can be continued at the original dose with close observation for worsening symptoms. For grade 2 events, DV should be held until recovery to grade ≤1 and then DV can be resumed at 1 dose level lower. For grade 3 events, at first occurrence DV should be held until recovery to grade ≤1, and then resumed at 2 dose levels lower than the original treatment dose. On a second occurrence of grade 3 events, DV should be discontinued.
If peripheral sensory symptoms/neuropathy does not recover enough to resume treatment within 28 days, then DV should be permanently discontinued. Counsel patients to report new numbness/tingling, balance issues, or fine‑motor changes promptly to more quickly mitigate peripheral sensory symptoms/neuropathy and hopefully allow patients to remain on therapy long-term.9,10
Disitamab Vedotin and Gastrointestinal Effects
GI effects such as nausea/vomiting, diarrhea/constipation can also occur with DV and are among the common AEs in clinical trials.10 However, DV is not classified as highly emetogenic and no antiemetic prophylaxis is mandated. In practice, providing risk‑adapted antiemetics, a take‑home as-needed therapy, and early loperamide guidance if diarrhea develops can help mitigate any severe GI symptoms, and supportive care can be escalated per local supportive care guidelines.9
Key Takeaways: Managing AEs Associated With HER2-Targeted ADC for GI, GU, and GYN Cancers
T‑DXd is a highly active HER2‑directed ADC now approved across multiple indications (including tumor‑agnostic IHC 3+ solid tumors). As with most ADCs, common AEs include many that are associated with standard chemotherapy; however, one main safety consideration with T-DXd is ILD/pneumonitis. It is important to educate patients on the signs and symptoms of ILD and to monitor for respiratory symptoms throughout treatment. Any symptoms or radiologic indications for ILD should prompt immediate investigation for ILD and if confirmed, interrupt T-DXd for grade 1 ILD and permanently discontinue T-DXd for grade ≥2 ILD. In addition to ILD, it is recommended to use prophylactic antiemetics to mitigate any nausea or vomiting, and CBC and LVEF levels should be assessed at baseline and during treatment to promptly identify and manage any instances of cytopenias or cardiac issues. For these AEs, follow the dose hold and dose reduction schedule for T-DXd according to the approved prescribing information instructions and do not reescalate T-DXd to maintain therapy safely and optimize patient outcomes.
DV is a HER2‑targeted MMAE ADC approved in China (gastric/GEJ and urothelial cancers, and as of May 2025 for HER2‑positive breast cancer with liver metastases). Day‑to‑day management of patients receiving DV centers on monitoring for and using dose holds and/or dose reductions for peripheral neuropathy, myelosuppression, transaminase elevations, and infusion reactions.
ADCs are becoming a more common class of therapy for many patients with cancer. Understanding the unique AE profile for each ADC and knowing the appropriate management strategies for these AEs can help to optimize patient outcomes by allowing patients to remain on therapy for the greatest efficacy while mitigating any AEs and maintaining patient quality of life.